2026-02-12 自然科学研究機構生命創成探究センター
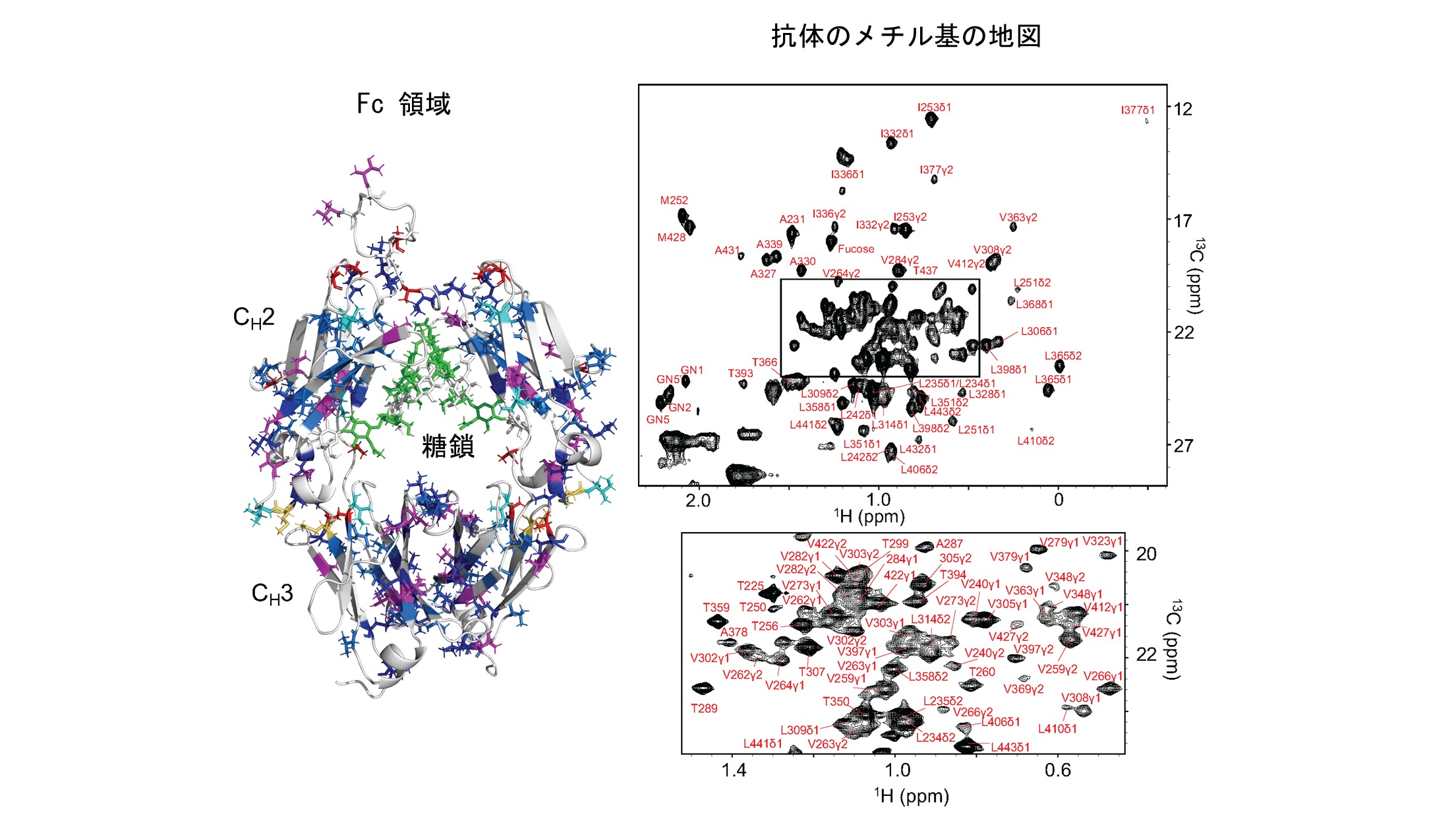
抗体のFc領域に存在するメチル基のNMR信号の地図
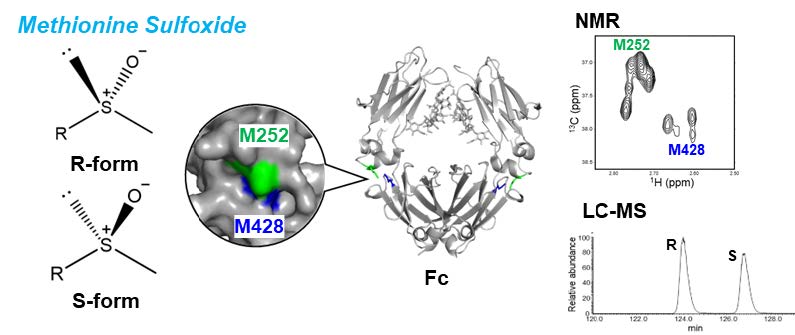
左:メチル基を含む残基を3次元モデル上に示している。アミノ酸残基タイプ別に色分けしている。右:抗体のFc領域のNMR信号およびその帰属
<関連情報>
IgG1 Fc領域の構造評価のための部位特異的メチル化を伴う非標識NMRアプローチ Unlabeled NMR Approach with Site-Specific Methyl Assignments for Structural Evaluation of the IgG1 Fc Region
Saeko Yanaka,Yuuki Koseki,Yohei Miyanoiri,Toshio Yamazaki,Tsutomu Terauchi,Daichi Kaneko,Yukiko Isono,Kohei Tomita,Sachiko Kondo,Masayoshi Onitsuka,Maho Yagi-Utsumi,Hirokazu Yagi,Akiko Ishii-Watabe,and Koichi Kato
Journal of the American chemical Society Published: February 11, 2026
DOI:https://doi.org/10.1021/jacs.5c18997
Abstract
Monoclonal antibodies are the cornerstone biopharmaceuticals whose safety and efficacy critically depend on their higher-order structure (HOS). Nuclear magnetic resonance (NMR) spectroscopy has emerged as a promising tool for HOS evaluation, yet its application has largely relied on fingerprinting approaches without residue-level interpretation. Here, we report site-specific assignments of methyl resonances in the Fc region of human IgG1, established through amino acid-selective labeling and correlation with backbone resonances using scalar coupling and NOE connectivities, further supported by mutagenesis. These assignments allowed us to identify glycoform-dependent spectral variations, including distinct signatures of core fucosylation and terminal galactosylation, as well as an Fc-specific amino acid substitution. Importantly, these spectral probes were detectable even in antibodies at natural isotopic abundance, enabling practical applications to therapeutic products without isotopic labeling. Furthermore, dynamic filtering highlighted methyl resonances from hinge and receptor-binding residues with elevated mobility, providing localized insights into functional sites. Collectively, our results establish unlabeled methyl NMR as a robust platform for sensitive and practical HOS assessment of therapeutic antibodies. This approach is broadly applicable to monitor glycosylation heterogeneity, chemical modifications, and batch-to-batch consistency, thereby offering a valuable framework for development and quality control of both innovative biopharmaceuticals and biosimilars.