2026-03-25 清華大学
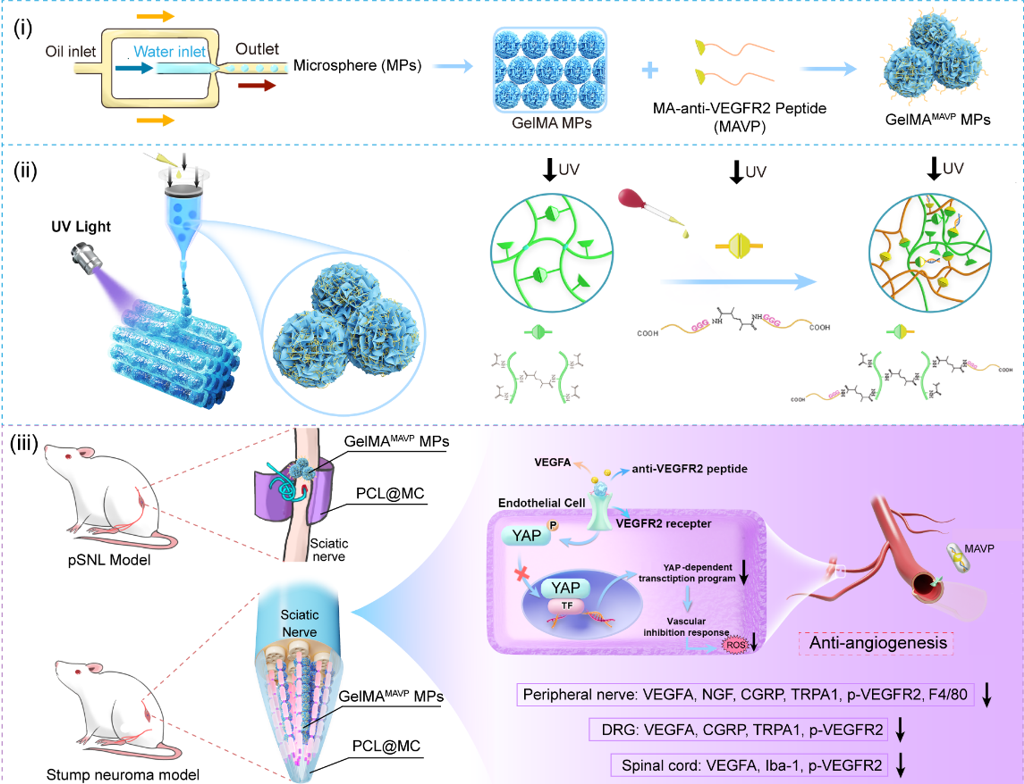
Figure 1. Fabrication of GelMAMAVP MPs using microfluidics and bioprinting technology to continuously regulate the residual nerve microenvironment and improve pain by targeting VEGFR2 antagonism.
<関連情報>
- https://www.tsinghua.edu.cn/en/info/1245/14750.htm
- https://www.sciencedirect.com/science/article/pii/S2452199X26001398
空間的に限定された導管内でVEGFR2阻害を標的とすることで、神経の自己修復が促進され、機械的アロディニアが軽減される Targeting VEGFR2 inhibition within a spatially-confined conduit promotes nerve self-resolution and alleviates mechanical allodynia
Xiaobin Luo, Toshitatsu Nagayasu, Yuhao Zhang, Zhi He, Jia Yang, Chengli Li, Guman Duan, Qunchuan Zong, Yun Liu, Kaiyuan Yang, Kaige Mao, Pengchao Ma, Zifan Li, Yating Zhao, KunKoo Kim, Jie Zhang, Zide Wang, Yuyang Luo, Jingsong Lu, Sengpav Tong…Xiumei Wang
Bioactive Materials Available online: 4 March 2026
DOI:https://doi.org/10.1016/j.bioactmat.2026.03.009
Highlights
- This platform facilitated the self-resolution of the nerve end interface, including sustained suppression of VEGFR2 phosphorylation and coordinated mechanotransduction signaling.
- Utilizes 3D bioprinting to pre-pattern GelMA MPs, resulting in aligned hydrogel microsphere fibers with topological structures.
- GelMAMAVP MPs distributed in the peripheral nerve stumps indirectly downregulated pain-related proteins in DRG and spinal horn cord.
Abstract
Stump neuromas represent nonfunctional redundant tissue that frequently elicits neuropathic pain through disorganized axonal regeneration, pathological angiogenesis, scar hyperplasia, and chronic neuroinflammation. Current therapeutic strategies fail to adequately address the underlying pathophysiology, particularly the excessive production of reactive oxygen species resulting from dysregulated vascular proliferation and activation. To tackle this issue, we developed photo-crosslinked gelatin methacryloyl (GelMA) microspheres covalently functionalized with methacryloyl-modified anti-VEGFR2 peptides (MAVP) to modulate pathological angiogenesis. These functionalized microspheres were fabricated into linear arrays via 3D printing and integrated within a progressively spatial-constrictive conduit. This platform facilitated the self-resolution of the nerve end interface, including sustained suppression of VEGFR2 phosphorylation and coordinated mechanotransduction signaling, thereby inhibiting neuroinflammatory responses and angiogenesis. In a sciatic nerve ligation model, GelMAMAVP MPs system demonstrated markedly superior analgesic efficacy over the conventional VEGFR2 inhibitor vandetanib. In a stump neuroma model, GelMAMAVP MPs effectively normalized the peripheral end interface microenvironment by inhibiting neovascularization, M1 macrophage polarization, and fibrotic scar formation. Furthermore, GelMAMAVP MPs distributed in the peripheral nerve stumps indirectly downregulated pain-related proteins (TRPA1/CGRP) in dorsal root ganglia and suppressed spinal microglial activation. Overall, this study presents a comprehensive and safe vascular-targeted strategy promoting nerve end interface self-resolution and prevention of neuropathic pain.