2026-03-25 浙江大学(ZJU)
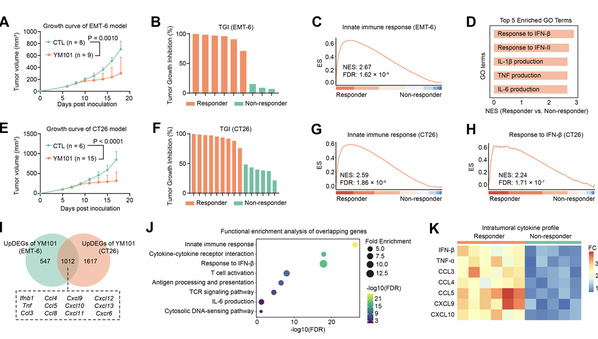
Figure 1. Distinct innate immune activation patterns are associated with differential response to YM101 treatment.
<関連情報>
- https://www.zju.edu.cn/english/2026/0325/c19573a3144257/page.psp
- https://www.nature.com/articles/s41467-026-69456-3
STING、TGF-β、PD-L1の三重標的化によりCXCL16–CXCR6シグナル伝達が増強され、強力な抗腫瘍応答が得られる Triple targeting of STING, TGF-β, and PD-L1 boosts CXCL16–CXCR6 signaling for potent antitumor response
Ming Yi,Tianye Li,Yinhui Gu,Mengke Niu,Dixuan Xue,Shengtao Hu,Yuze Wu,Bin Zhao,Di Zhang,Yingkang Ma,Minjun Zhang,Jing Zhang,Yongxiang Yan,Pengfei Zhou,Xiaojun Zhang,Zhuxian Zhou,Qian Chu,Kongming Wu & Zhijun Dai
Nature Communications Publoished:09 February 2026
DOI:https://doi.org/10.1038/s41467-026-69456-3
Abstract
Antibodies targeting TGF-β and PD-L1 initially showed promise as second-generation PD-L1 agents. However, consecutive trial failures have limited their clinical success. Our study reveals that the efficacy of the TGF-β×PD-L1 bispecific antibody (BsAb) is compromised by insufficient activation of innate immune responses. To address this, we combine STING agonists with the BsAb, significantly enhancing tumor suppression beyond that achieved with standard STING agonist plus anti-PD-L1 combinations in preclinical models. Unexpectedly, even STING agonist monotherapy is improved by TGF-β blockade, suggesting that TGF-β suppresses STING-driven immune activation. We find that this synergy is mediated by the CXCL16–CXCR6 axis, where STING activation and TGF-β blockade promote CXCL16 expression in macrophages and dendritic cells, recruiting and sustaining cytotoxic CXCR6+ T cells. Additionally, PD-L1 blockade further enhances their antitumor activity. To optimize this strategy, we develop Y101S, an antibody-drug conjugate targeting TGF-β, PD-L1, and STING, which demonstrates superior tumor control and immune modulation in preclinical models. These findings highlight the therapeutic potential of this triple-targeting approach.