2026-04-02 中国科学院(CAS)
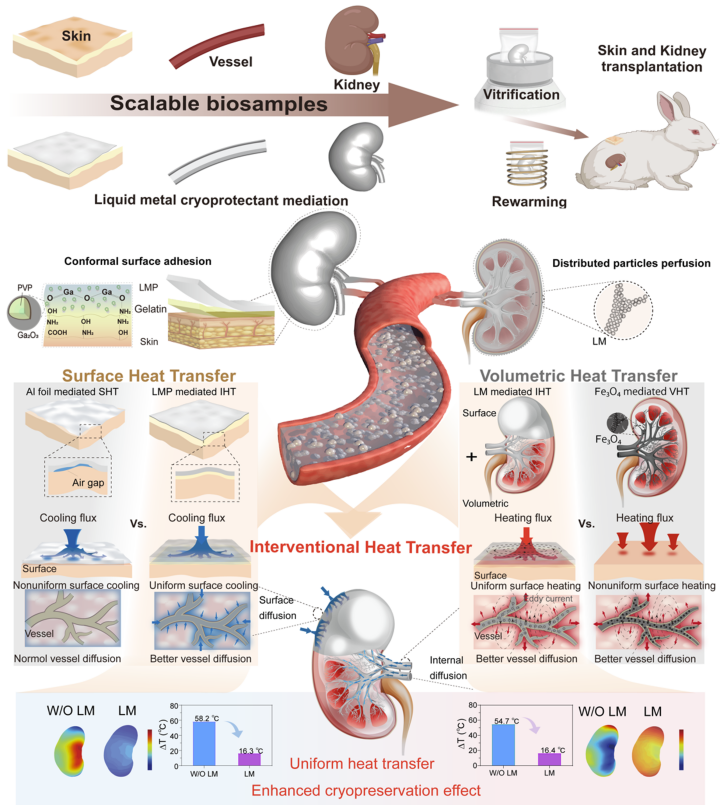
Liquid metal cryoprotectants mediated multi-scale interventional enhanced heat transfer. (Image by TIPC)
<関連情報>
- https://english.cas.cn/newsroom/research-news/202604/t20260402_1155069.shtml
- https://www.cell.com/matter/abstract/S2590-2385(26)00127-X
生体試料の大規模凍結保存のための液体金属凍結保護剤 Liquid metal cryoprotectants for scalable biosamples cryopreservation
Yi Hou ∙ Xiaohong Wang ∙ Jie Zhang, ∙ … ∙ Xiaoying Li ∙ Jing Liu ∙ Wei Rao
Matter Published:March 30, 2026
DOI:https://doi.org/10.1016/j.matt.2026.102764
Progress and potential
Cryopreservation is currently the most efficient way for long-term preservation of biological resources. Especially, the cryopreservation of large-sized biosamples has tremendous value in tissue engineering and organ transplantation. The core issue lies in the low thermal conductivity of biosamples, which leads to bottlenecks that restrict sufficient heat transfer during cooling and rewarming. In this work, we overcome this critical barrier by introducing a high thermal conductivity liquid metal (LM) cryoprotectant and interventional heat transfer (IHT) strategy. By coating tissues with or perfusing blood vessels with LM cryoprotectants, we drastically reduce interfacial and contact thermal resistance and boost warming rates. This approach acts as a “thermal highway” throughout the tissue, ensuring fast and uniform heat transfer that cuts thermal stress by a 100-fold. We successfully demonstrated this method on scalable biological samples, including rabbit skins, arteries, and kidneys. Remarkably, this technique enabled the first successful allogeneic transplantation of a vitrified rabbit kidney, supporting the survival of the recipient. This breakthrough provides a universal, multiscale heat transfer solution for the conformal cryopreservation of complex tissues. By breaking up the thermal barrier restricting the cryopreservation of scalable biosamples, our methodology paves the way for the establishment of large-scale organ banks. Ultimately, this could relieve the severe shortage of donor organs and revolutionize life-saving transplantation surgeries worldwide.
Highlights
- LM cryoprotectant owns the highest conductivity among the cryoprotectant media
- IHT ensures uniform heat transfer and cuts thermal stress by a 100-fold
- IHT improves the viability of scalable biosamples including skin, vessel, and kidney
- IHT enabled the first allogeneic transplantation of rabbit kidney after vitrification
Summary
Cryopreservation of scalable biosamples remains a long-standing biomedical target yet is hindered by inefficient heat transfer causing irreversible thermal stress damage. Here, we propose a liquid metal (LM) cryoprotectant combined with an interventional heat transfer (IHT) methodology to synergistically reduce the interfacial thermal resistance and accelerate warming rates. This principle integrates LM-based high thermal conductivity media (9.3 W/m·K, 10.3 times higher than that of cryoprotectants containing Fe3O4 nanoparticles in nanowarming) with surface and vascularized perfusion. The IHT efficacy is evidenced by 1.7-fold and 3.6-fold increases in skin and arterial viability, compared with conventional water bath controls, alongside the strong self-healing capability of transplanted skin after cryopreservation. Notably, the rewarmed kidneys showed excellent morphological and functional maintenance, enabling the first successful allogeneic transplantation and recipient survival in rabbits. Our work opens a multiscale enhanced heat transfer paradigm based on LM cryoprotectants for conformal cryopreservation, offering transformative potential for large-scale organ banking and transplantation.