2026-04-08 合肥物質科学研究院(HFIPS)
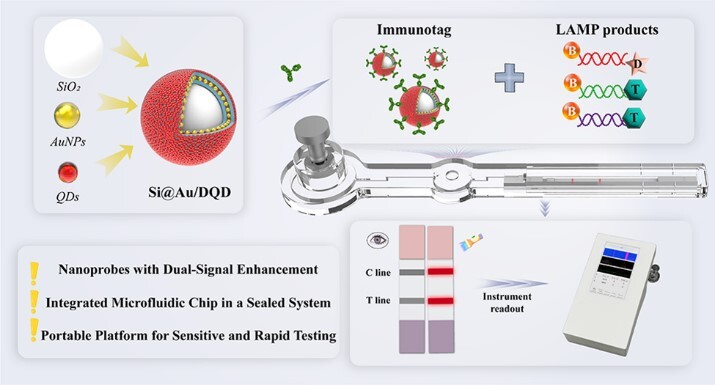
Integrated “Isothermal Amplification–Immunochromatography” Detection Platform (Image by DAI Liping)
<関連情報>
- https://english.hf.cas.cn/nr/rn/202604/t20260408_1155386.html
- https://pubs.acs.org/doi/10.1021/acs.analchem.5c05437
医療現場における医療関連感染病原体の高感度検出のための、デュアルシグナルナノプローブを備えたプッシュボタン式マイクロ流体プラットフォーム Push-Button Microfluidic Platform with Dual-Signal Nanoprobes for Enhanced Sensitivity Detection of Healthcare-Associated Infection Pathogens at Point-of-Care
Liping Dai,Yun Wang,Xiaosong Wu,Qian Wang,Zhigang Li,Yong Liu,Long Zhang,Shuai Zheng,and Shu Wang
Analytical Chemistry Published: February 9, 2026
DOI:https://doi.org/10.1021/acs.analchem.5c05437
Abstract
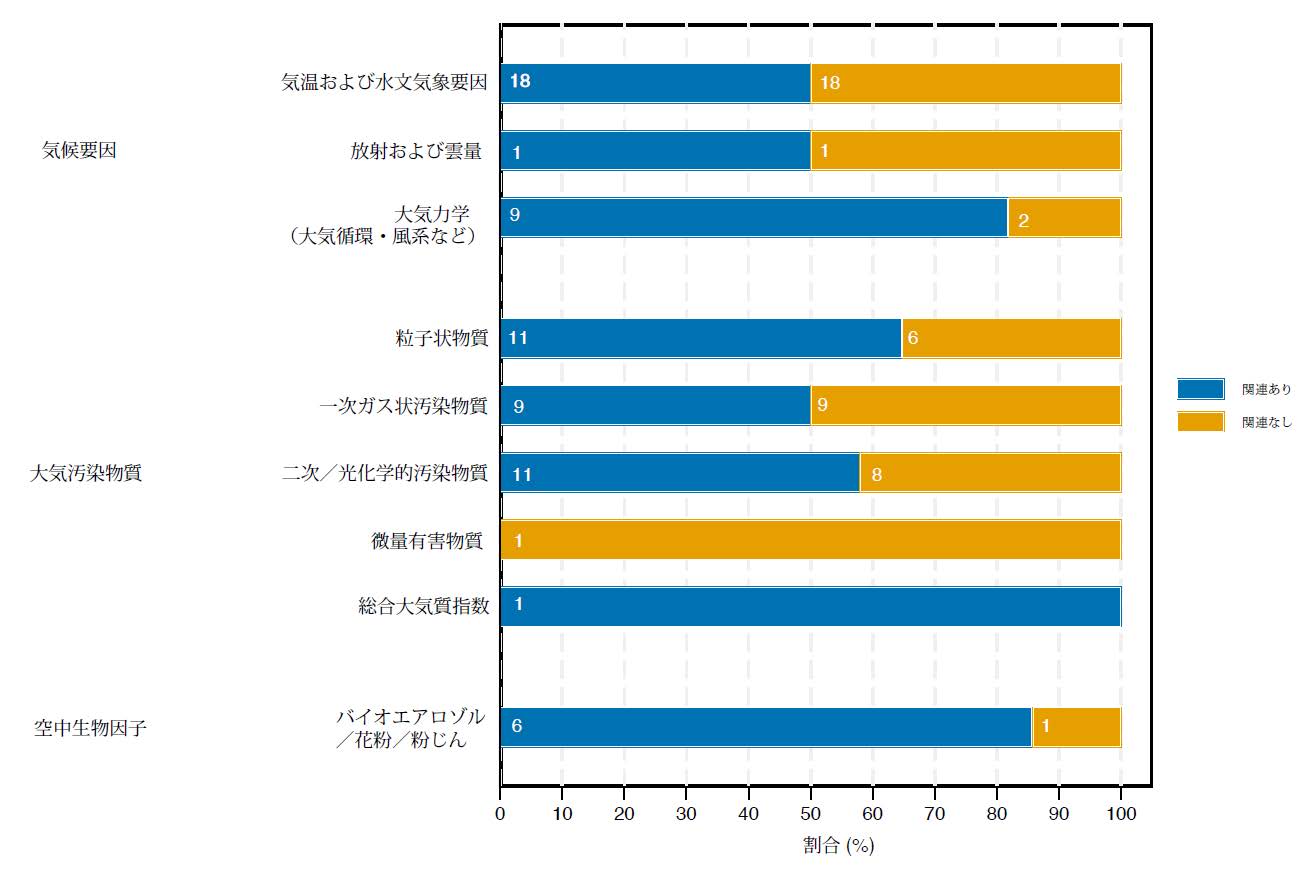
Healthcare-associated infection (HAI) pathogens cause severe nosocomial outbreaks, jeopardizing patient safety and straining healthcare systems. Conventional loop-mediated isothermal amplification–lateral flow immunoassay (LAMP-ICA) allows rapid pathogen detection but is constrained by low sensitivity, a high false-positive rate, and an extended detection time. To address these limitations, we present a dual-mode (colorimetric/fluorescent) microfluidic biosensing platform based on silicon–gold/quantum dot core–shell nanoprobes (Si@Au/DQD NPs). The platform incorporates two key innovations: (1) The colorimetric/fluorescent dual-signal Si@Au/DQD nanoprobe enhances detection reliability and sensitivity through dual-signal complementary verification and multilayered QD design, halving the LAMP amplification time compared to traditional colloidal gold systems, and (2) a modular microfluidic chip integrates LAMP amplification and ICA detection within a closed system, effectively preventing leakage and contamination of amplification products. Performance evaluation showed that the fluorescence detection limit of this system for Staphylococcus aureus (S. aureus), Legionella pneumophila (L. pneumophila), and Klebsiella pneumoniae (K. pneumoniae) reaches 82–140 CFU/mL, with the entire process completed within 30 min. In addition, the detection of 25 clinical environmental samples verifies the practicality of the designed integrated detection platform. With high sensitivity, strong specificity, and dual-mode capability for qualitative colorimetric screening and quantitative fluorescence analysis, this technology offers an efficient solution for point-of-care testing (POCT) of HAI pathogens, particularly in resource-limited settings and in on-site emergency diagnostics.