2026-04-08 エディンバラ大学
<関連情報>
- https://www.ed.ac.uk/news/crohns-disease-bowel-scarring-trigger-identified
- https://pathsocjournals.onlinelibrary.wiley.com/doi/10.1002/path.70019
クローン病のリンパ球凝集塊と内皮細胞クラスターは、線維狭窄型クローン病の粘膜下線維症と共局在する Crohn’s lymphoid aggregates with endothelial clusters colocalise with submucosal fibrosis in fibrostenosing Crohn’s disease
Michael Glinka, Gregory J Wickham, Francesca Nadalin, Kathryn J Kirkwood, Helen Caldwell, Mike Wicks, Bill Hill, Derek Houghton, Mehran Sharghi, Amirhosein Kefayat, Bernard Haggarty, …
The Journal of Pathology Published: 05 February 2026
DOI:https://doi.org/10.1002/path.70019

Abstract
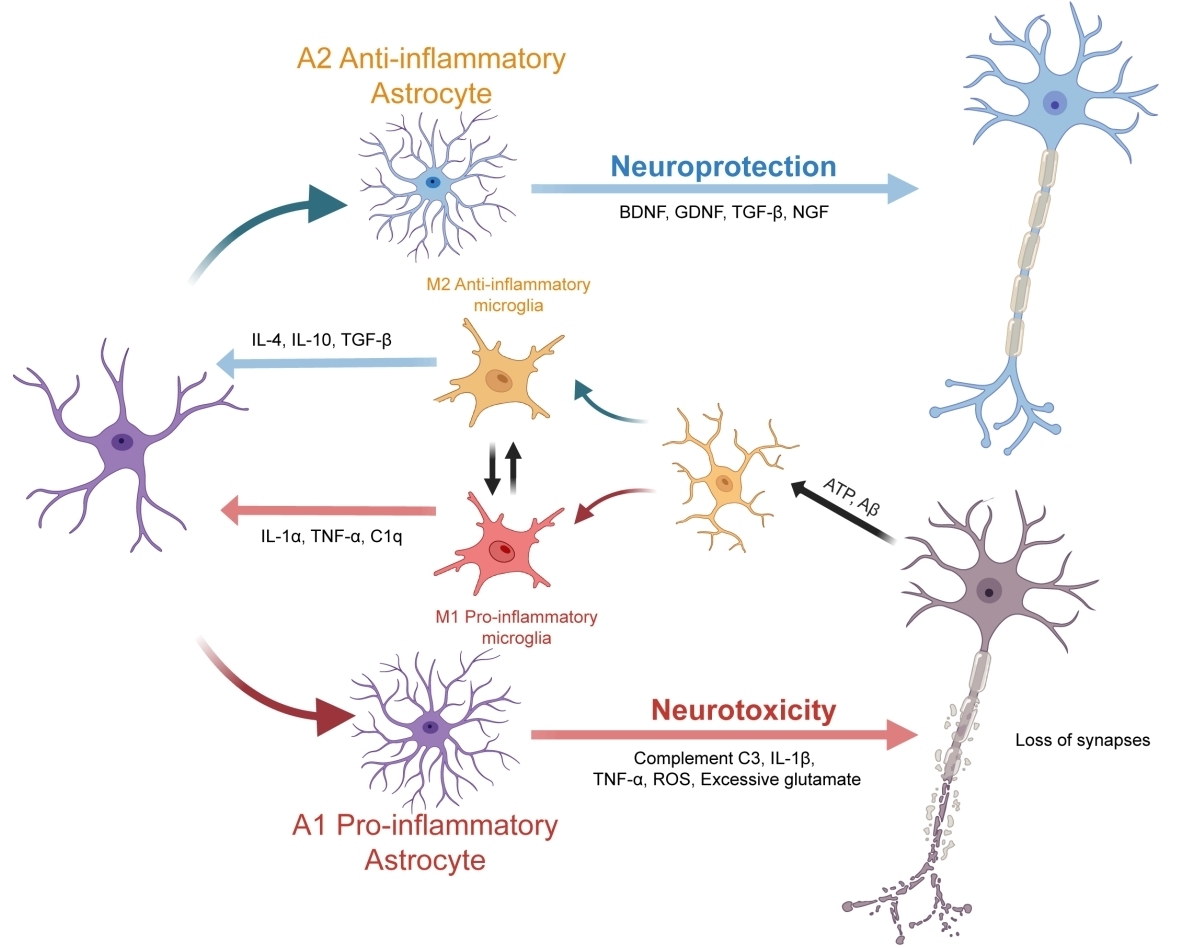
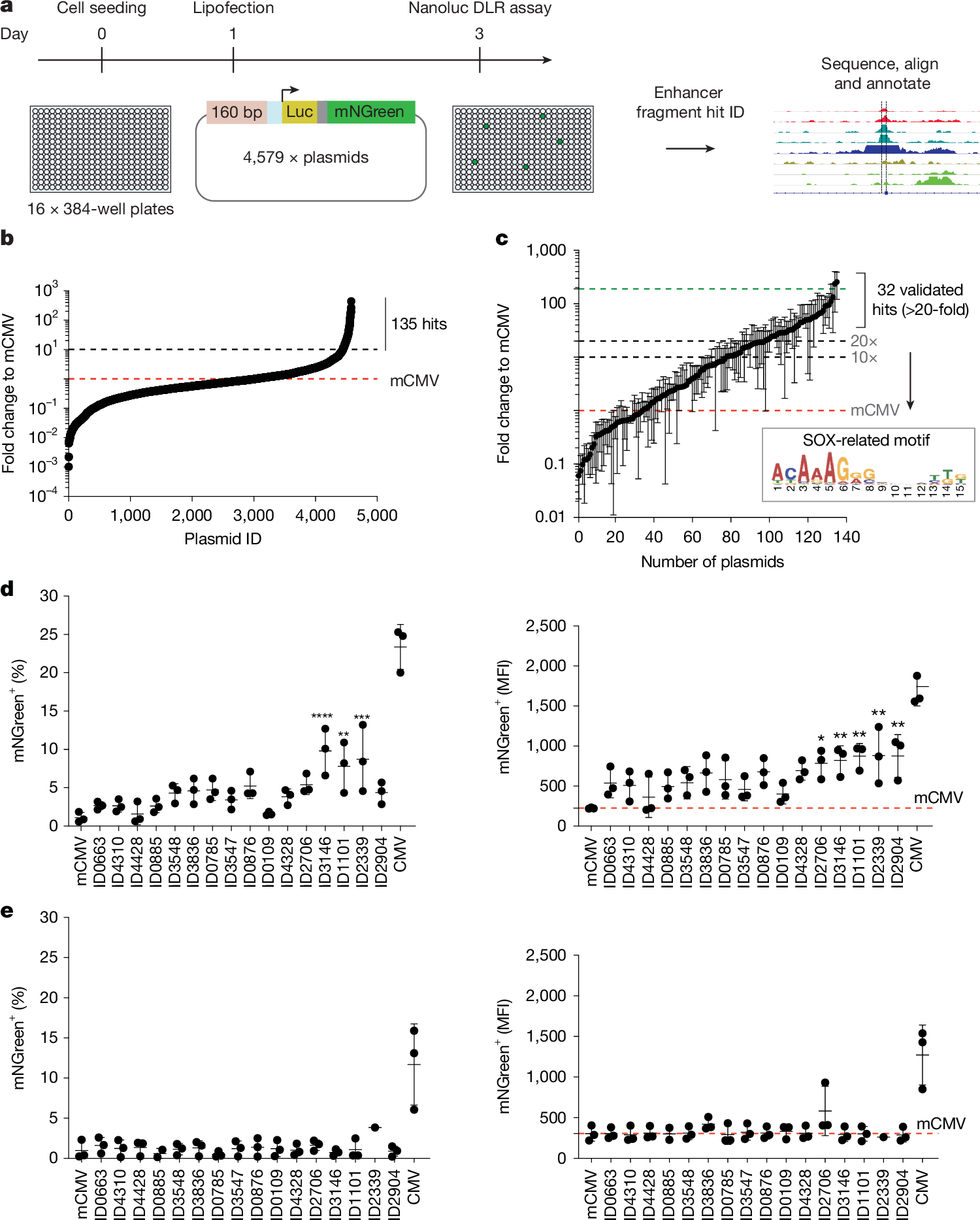
Crohn’s disease (CD) involves chronic transmural inflammation of the intestines, leading to progressive wall fibrosis with stenosis and luminal obstruction, predominantly in the terminal ileum. Fibrosis is a significant therapeutic challenge, thus improved understanding of localisation, cellular composition, and cell–cell interactions in CD fibrostenosing lesions (FSLs) may identify potential targetable pathways. Using CD FSL patient resection samples, we identify and quantify novel pathological changes in structure, collagen, and cell numbers for each ileal layer (mucosa, muscularis mucosae, submucosa, muscularis propria, serosa). In addition, fresh resection ileal samples were single-cell RNA (scRNA)-sequenced, validating the cell types and cell–cell interactions. We found significantly increased collagenous fibrosis expansion, significantly increased infiltration of lymphocytes, macrophages, endothelium, and Crohn’s lymphoid aggregates (CLAs) in all layers, except for the ulcerated mucosa. Importantly, endothelial cells accumulate in clusters around CLAs, and scRNA-seq data demonstrated ligand–receptor intercellular signalling interactions between endothelium, B and T lymphocytes, macrophages, and myofibroblasts via multiple pathways that included GAS, SELL, and SELPLG, among many others. The highest levels of fibrotic collagen and CLAs with accumulated endothelium were observed in submucosa, followed by serosa, demonstrating colocalisation and correlation of endothelial-CLAs with collagen that is consistent with CLAs having a role in promoting collagenous fibrosis that requires further investigation. © 2026 The Author(s). The Journal of Pathology published by John Wiley & Sons Ltd on behalf of The Pathological Society of Great Britain and Ireland.