2026-02-27 カリフォルニア大学アーバイン校(UCI)
<関連情報>
- https://news.uci.edu/2026/02/27/uc-irvine-scientists-uncover-cellular-mechanism-behind-muscle-repair/
- https://www.nature.com/articles/s42255-026-01457-4
PFKMは骨格筋分化における代謝変化を制御する PFKM governs metabolic shifts throughout skeletal muscle differentiation
Melissa Campos,Steven T. Nguyen,Xiangduo Kong,Ying Yang,Richard L. Watson,Anastasia Gromova,Catherine R. Livelo,Carolina N. Franco,Julia E. Cabral,Laurence J. Seabrook,Shengqi Dai,Yingzi Liu,Mingqi Zhou,Eric A. Hanse,Kaelyn Sumigray,Albert R. La Spada,Marcus M. Seldin,Maksim V. Plikus,Dequina A. Nicholas,Reginald McNulty,Mei Kong,Kyoko Yokomori & Lauren V. Albrecht
Nature Metabolism Published:24 February 2026
DOI:https://doi.org/10.1038/s42255-026-01457-4
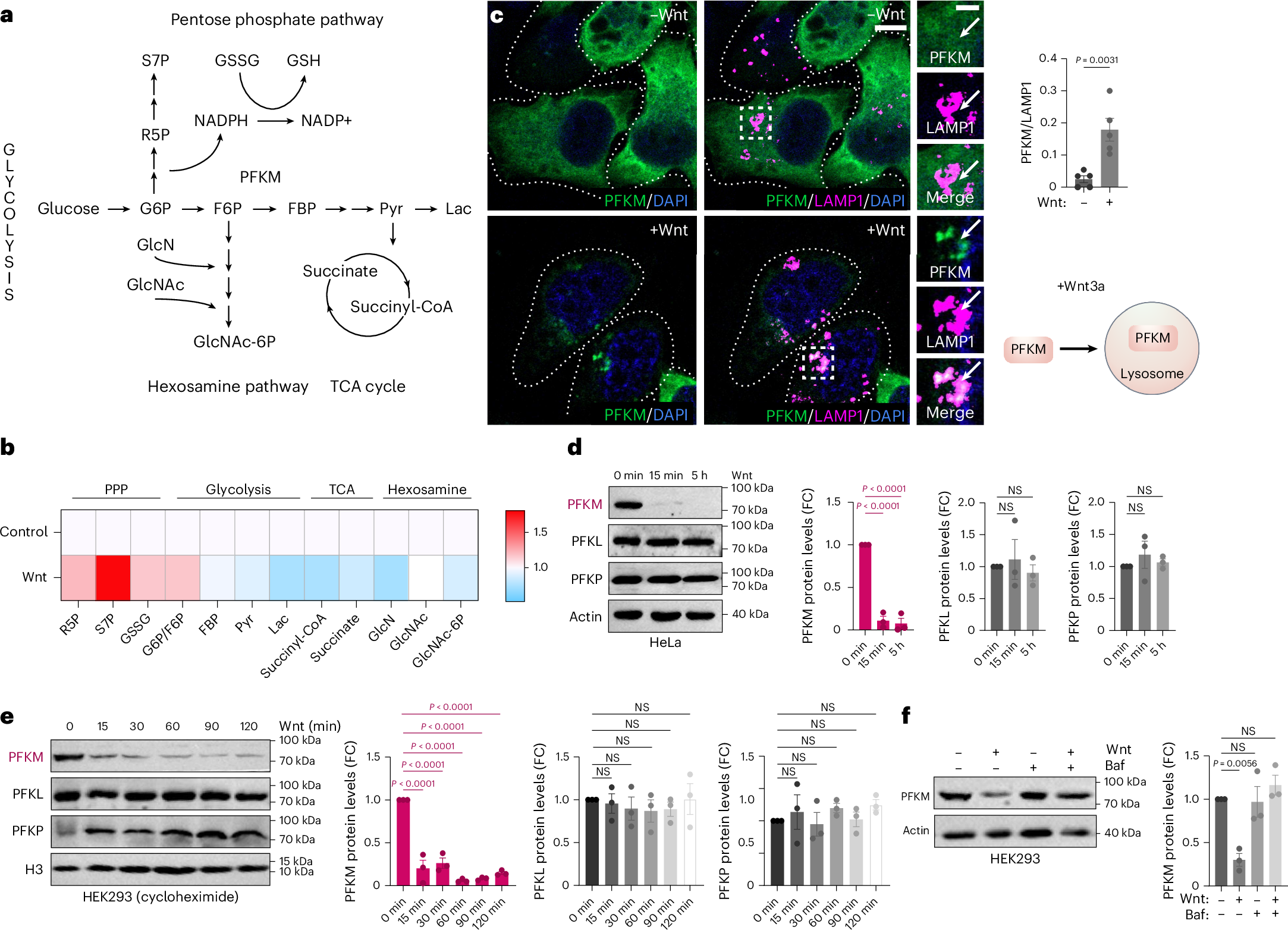
Abstract
Metabolism is known to influence cell identity, but the underlying mechanisms remain unclear. Here we reveal spatiotemporal dynamics of phosphofructokinase 1 (PFK1), a key glycolytic enzyme, within the skeletal muscle lineage. The expression of PFKM (the muscle isoform of PFK1) is low in muscle stem cells and increases during differentiation. Mechanistically, Wnt signalling rapidly induces lysosomal degradation of PFKM through a methyl arginine degron motif, which gets selectively methylated by the protein arginine methyltransferase (PRMT1) and delivered to lysosomes through microautophagy. PFKM degradation shifts glucose metabolism from glycolysis to the pentose phosphate pathway. PFKM overexpression increases glycolysis and promotes differentiation into terminally differentiated myofibres. On the other hand, PFKM knockdown blunts differentiation, which can be rescued by supplementation with the downstream glycolytic intermediate 3-phosphoglycerate. In sum, our findings highlight the importance of compartmentalized metabolism in cell fate decisions.


