2026-02-24 名古屋大学
<関連情報>
- https://www.nagoya-u.ac.jp/researchinfo/result/2026/02/alstdp-43.html
- https://www.nagoya-u.ac.jp/researchinfo/result/upload_images/20260224_med_jp.pdf
- https://www.pnas.org/doi/10.1073/pnas.2513642123
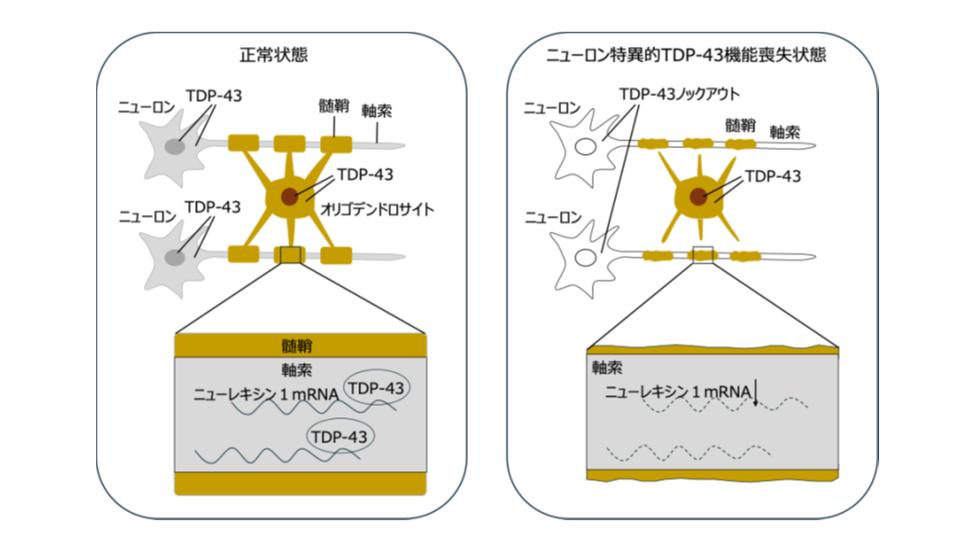
神経細胞のTDP-43はニューレキシン1 mRNAの安定化を介してミエリン形成を制御する Neuronal TDP-43 regulates myelin formation via neurexin 1 mRNA stabilization
Jiayi Li, Yohei Iguchi, Kenji Yoshida, +16 , and Masahisa Katsuno
Proceedings of the National Academy of Sciences Published:February 25, 2026
DOI:https://doi.org/10.1073/pnas.2513642123
Significance
TAR DNA-binding protein 43 (TDP-43) is a major component of the pathological inclusion of neurons of Amyotrophic lateral sclerosis and frontotemporal lobar degeneration, and dysfunction of TDP-43 has been thought to be a major cause of these diseases. This study demonstrates that loss of TDP-43 in neurons induces hypomyelination, and this impairment was rescued with the supplementation of cytoplasmic TDP-43 in neurons. Using fluorescence-activated cell sorting and RNA sequencing, we identified NRXN1 as the factor which promotes myelin formation and is regulated by TDP-43. Supplementation of NRXN1 in the neurons restored the hypomyelination in TDP-43cKO mice. These findings suggest the pivotal roles of neuronal TDP-43 in neuron–oligodendrocyte interactions.
Abstract
Amyotrophic lateral sclerosis (ALS) and frontotemporal lobar degeneration (FTLD) develop as spatial pathologies in which neurons and glial cells are interconnected. TAR DNA-binding protein 43 (TDP-43) is a major pathological protein that is inextricably associated with ALS and FTLD. In this study, we investigated the roles of neuronal TDP-43 in neuron–oligodendrocyte interactions using neuron-specific TDP-43 knockout (TDP-43cKO) mice. TDP-43 depletion in neurons induced hypomyelination, which was confirmed by immunohistochemistry and ultrastructural analysis. In addition, conduction disturbance was revealed by electrophysiological analysis. The hypomyelination of TDP-43cKO mouse was restored by cytoplasmic TDP-43 supplementation in neurons. Neuron-specific transcriptome analysis revealed that neurexin 1 (NRXN1) is the regulatory target of TDP-43, which promotes myelin formation. The hypomyelination of TDP-43cKO mice was also restored by NRXN1b supplementation in neurons. We further confirmed that TDP-43 stabilizes Nrxn1 mRNA by binding to the Nrxn1 3’untranslated region (3’UTR). Although TDP-43cKO exhibited impaired recognition memory, the supplementation of NRXN1 in the hippocampus recovered the memory disturbances. In conclusion, this study demonstrates the neuron–oligodendrocyte interaction mediated by neuronal TDP-43 via NRXN1 mRNA stabilization. These findings shed light on neuron–oligodendrocyte interaction in the disease mechanisms of ALS/FTLD.