2026-03-03 東京科学大学
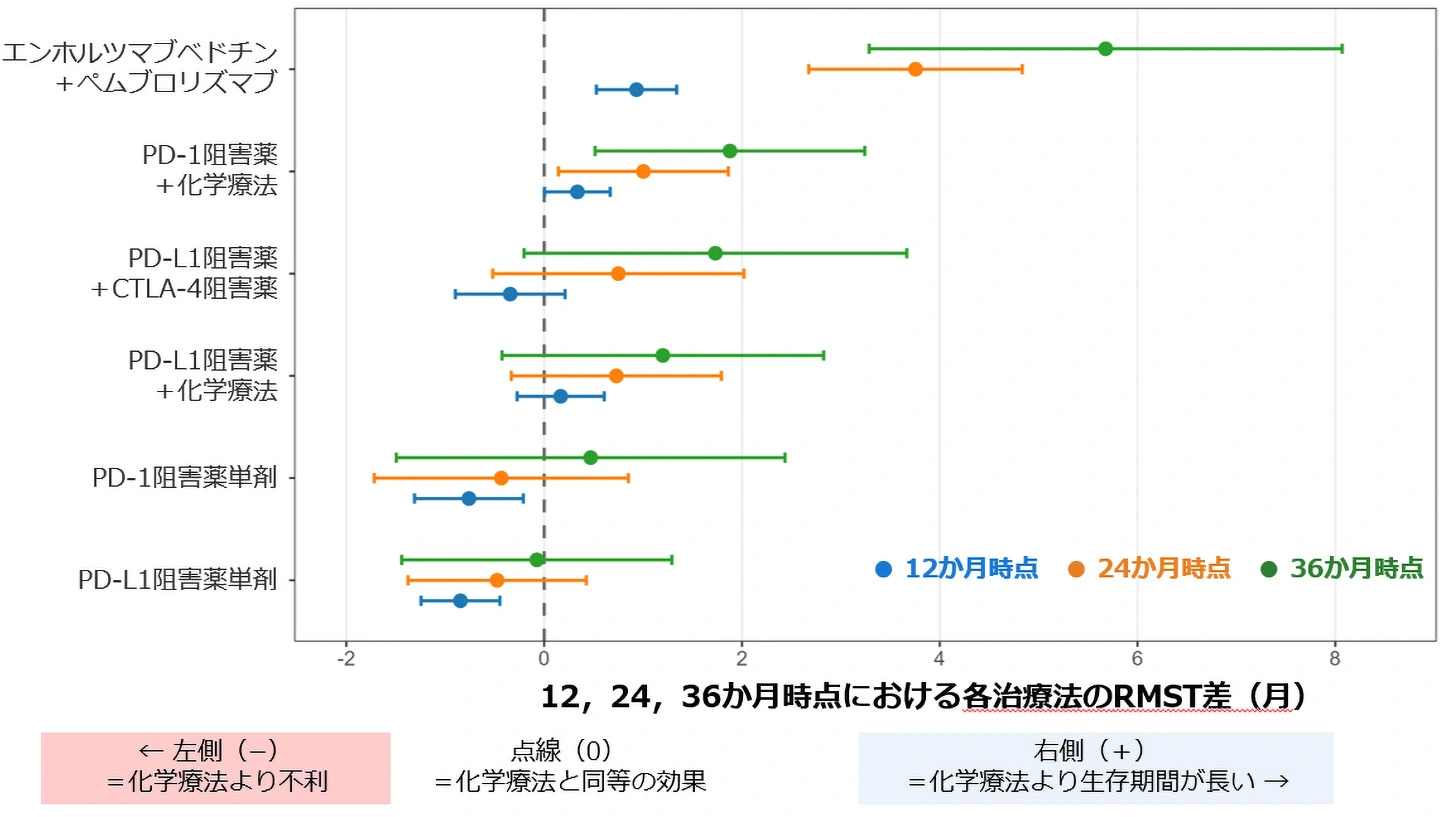
図1. 化学療法と比較した各治療法のRMST差
<関連情報>
- https://www.isct.ac.jp/ja/news/arv3145v3rd5#top
- https://www.sciencedirect.com/science/article/pii/S2666168326000200
制限平均生存期間を用いた進行尿路上皮癌の治療の再評価:系統的レビューとネットワークメタアナリシス Re-evaluating Treatments for Advanced Urothelial Carcinoma Using Restricted Mean Survival Time: A Systematic Review and Network Meta-analysis
Shugo Yajima, Wei Chen, Kohei Hirose, Akihiro Hirakawa, Kenji Tanabe, Motohiro Fujiwara, Hiroshi Fukushima, Hajime Tanaka, Hitoshi Masuda, Yasuhisa Fujii, Soichiro Yoshida
European Urology Open Science Available online: 6 February 2026
DOI:https://doi.org/10.1016/j.euros.2026.01.013
Abstract
Background and objective
The proportional hazards (PH) assumption may not hold for immune checkpoint inhibitor (ICI) trials because of delayed treatment effects. We assessed PH validity in trials of first-line treatment in advanced urothelial carcinoma (UC) and compared treatments using restricted mean survival time (RMST).
Methods
We conducted a systematic review and network meta-analysis (NMA) of phase 2/3 randomized controlled trials published between 2015 and 2025 evaluating ICIs or antibody-drug conjugates. Seven trials involving 5321 patients with ten treatment comparisons were analyzed. Individual patient data were reconstructed from Kaplan-Meier curves. The PH assumption was tested using Schoenfeld residuals (p < 0.05 indicating violation). RMST-based NMA was performed at 6-mo intervals up to 36 mo.
Key findings and limitations
The PH assumption was violated in 50% of overall survival (OS) comparisons and 60% of progression-free survival comparisons. ICI monotherapy regimens demonstrated a significant early detriment at 12 mo in comparison to chemotherapy (PD-1 inhibitors: −0.8 mo, p = 0.007; PD-L1 inhibitors: −0.9 mo, p < 0.001) that was completely masked by nonsignificant hazard ratios. Enfortumab vedotin + pembrolizumab was associated with a superior RMST benefit of 5.7 mo (95% confidence interval 3.3–8.1) at the 36-mo time point. Study limitations include the sparse network structure and heterogeneous patient eligibility criteria across trials.
Conclusions and clinical implications
Half of modern UC trials violate PH assumptions, with time-dependent treatment effects that can be obscured by traditional analyses. RMST analysis quantifies the magnitude and clinical impact of early ICI monotherapy detriment that is not captured by nonsignificant hazard ratios. Enfortumab vedotin plus pembrolizumab demonstrated the largest RMST benefit in this analysis, although this finding is based on a single pivotal trial and requires validation in additional studies and real-world settings. These findings support the value of RMST analysis as a complementary approach for evaluating time-dependent treatment effects in immunotherapy trials.
Patient summary
We analyzed clinical trials of treatments for advanced bladder cancer and found that traditional statistical methods may miss important treatment patterns. Our analysis shows that immunotherapy alone may perform worse than chemotherapy initially, but the treatment effect improves over time. The combination of enfortumab vedotin and pembrolizumab provides the best outcomes for patients at all time points.