2026-03-10 カリフォルニア大学サンディエゴ校(UCSD)
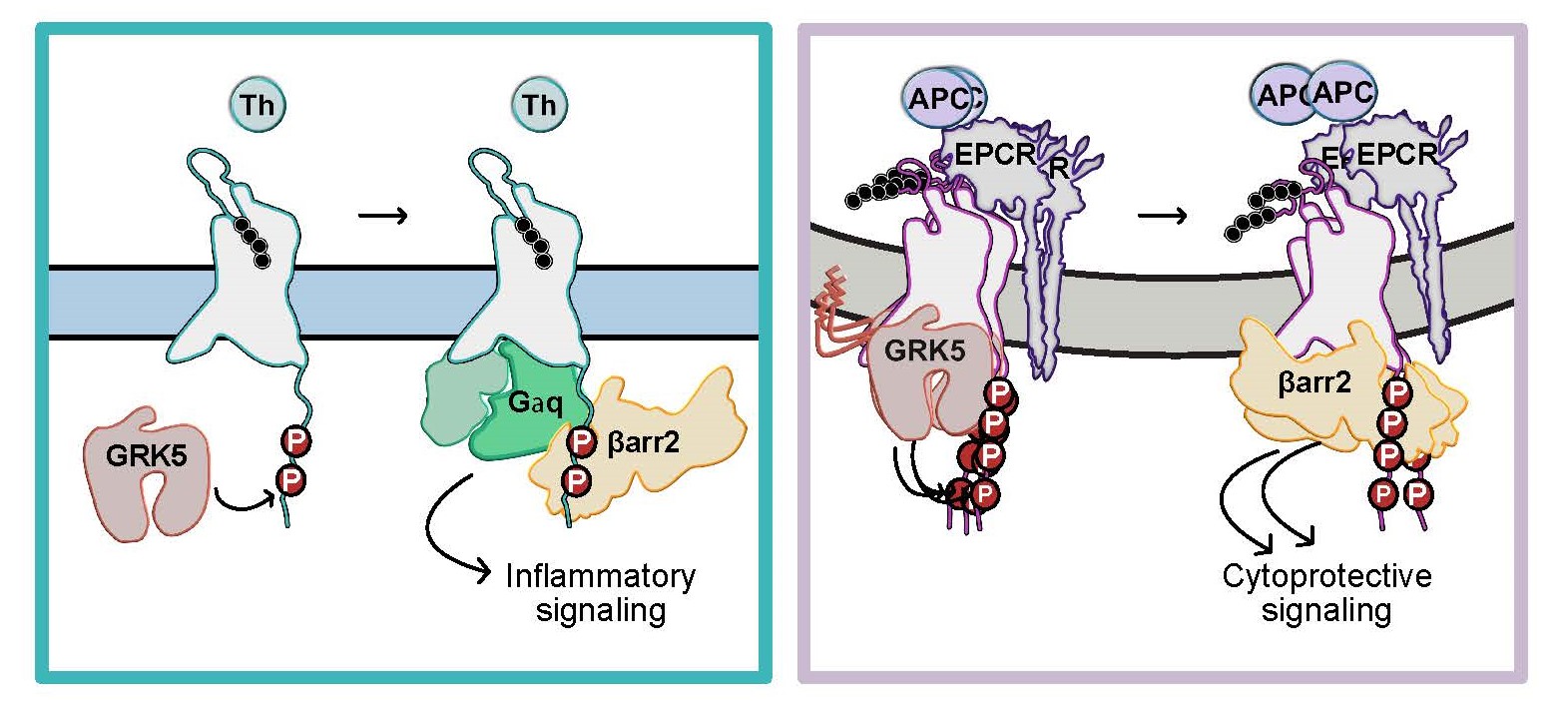
This diagram illustrates the two different responses triggered by PAR1. PAR1 can trigger inflammation when activated by thrombin (left) or protection when activated by APC (right). GRK5 and β-arrestin2 each play key roles in these processes, but in different ways depending on the activator.
<関連情報>
- https://today.ucsd.edu/story/how-one-receptor-can-help-or-hurt-your-blood-vessels
- https://www.cell.com/cell-reports/fulltext/S2211-1247(26)00119-1
プロテアーゼ活性化受容体1のシグナル伝達バイアスは、GRK5とβアレスチン2の異なる決定因子によって決定される Signaling bias of the protease-activated receptor-1 is dictated by distinct GRK5 and β-arrestin-2 determinants
Monica L. Gonzalez Ramirez ∙ Lennis B. Orduña-Castillo ∙ Carolyne Bardeleben ∙ … ∙ Cierra A. Birch ∙ Irina Kufareva ∙ JoAnn Trejo
Cell Reports Published:March 3, 2026
DOI:https://doi.org/10.1016/j.celrep.2026.117041
Highlights
- AlphaFold 3 modeling explains the basis of PAR1 signaling bias to thrombin and APC
- GRK5 membrane anchoring differentially regulates thrombin and APC signaling bias
- PAR1 phosphorylation and β-arrestin binding distinguish thrombin versus APC signaling
- Unique β-arrestin conformations underlie thrombin versus APC bias signaling
Summary
G protein-coupled receptors (GPCRs) exhibit signaling bias or preferential activation of heterotrimeric G proteins versus GPCR kinase (GRK)-mediated β-arrestin signaling. The protease-activated receptor-1 (PAR1) activates both G protein and β-arrestin in response to thrombin but only β-arrestin in response to activated protein C (APC). Thrombin-activated PAR1-G protein signaling is desensitized by β-arrestin-1, whereas APC-activated PAR1 signaling is propagated by β-arrestin-2. The mechanisms underlying PAR1 biased signaling are not known. Here, using computational modeling combined with cellular and biochemical studies, we reveal the molecular basis of signaling by thrombin- and APC-activated PAR1. Although both thrombin- and APC-induced PAR1 signaling are regulated by the same GRK, GRK5, the two types of signaling are differentially dependent on GRK5 membrane anchoring, PAR1 C-terminal phosphorylation sites, and the binding mode of β-arrestin-2. These differences translate into distinct β-arrestin-2 conformations and define the APC cytoprotective signaling signature, which contrasts with thrombin inflammatory signaling.

00006-X/asset/b90eca0d-b03e-45c5-a8f5-a91f0b4c41be/main.assets/gr1_lrg.jpg)
