2026-03-12 東京大学,明治大学,京都大学,東北大学,金沢大学,筑波大学,科学技術振興機構
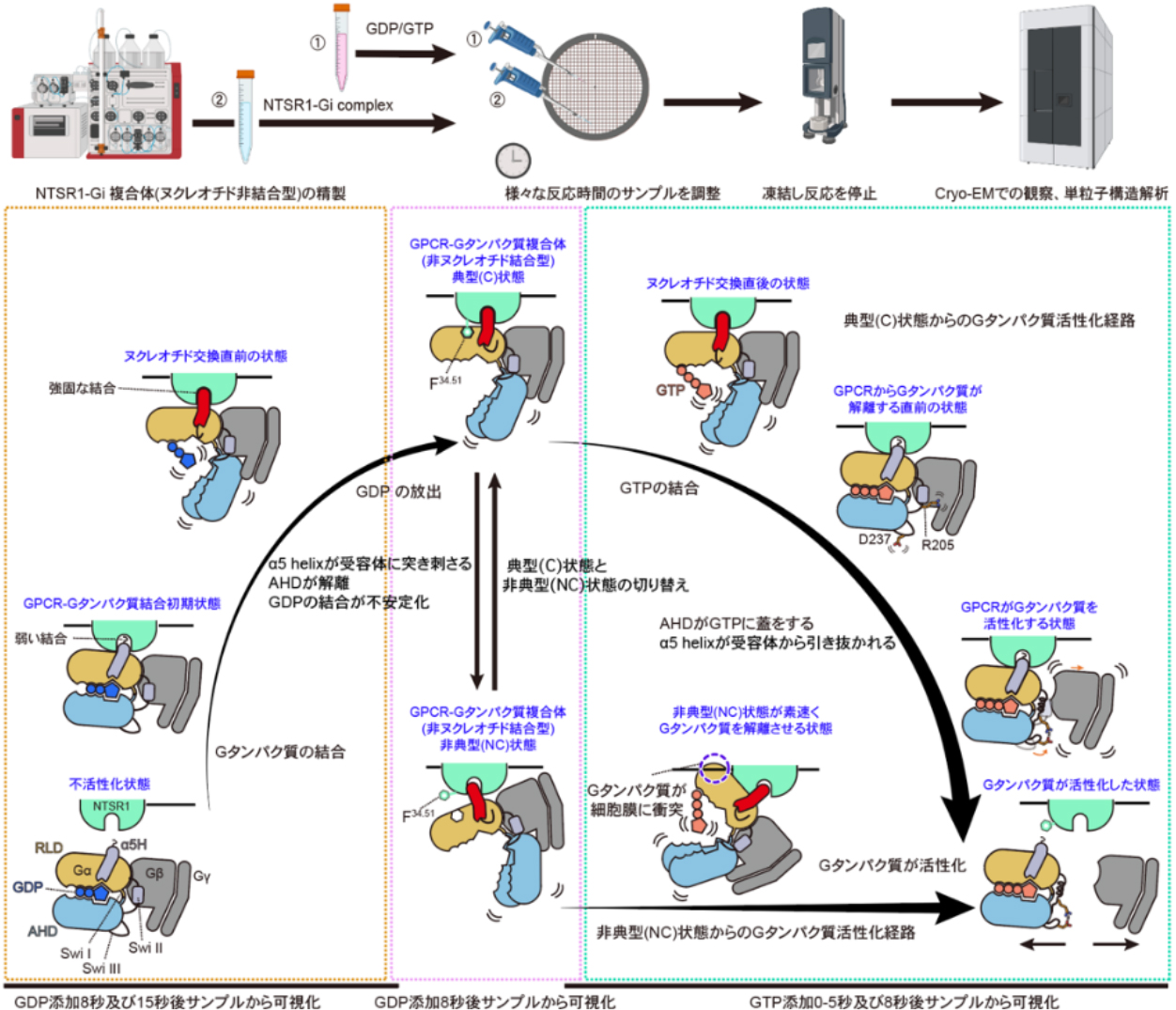
本研究の概略図
<関連情報>
- https://www.rcast.u-tokyo.ac.jp/ja/news/release/20260312.html
- https://www.nature.com/articles/s41586-026-10228-w
GPCRによるGタンパク質の認識と活性化の動的基盤 The dynamic basis of G-protein recognition and activation by a GPCR
Kazuhiro Kobayashi,Kouki Kawakami,Toshiki E. Matsui,Shun Yokoi,Masahiro Fukuda,Tomohiro J. Narita,Hiroki Arai,Mai Tambo,Takashi Sumikama,Manae Tatsumi,Keitaro Yamashita,Junki Koyanagi,Mai Kugawa,Hisako Ikeda,Ayumi Sumino,Ayori Mitsutake,Brian K. Kobilka,Asuka Inoue & Hideaki E. Kato
Nature Published:11 March 2026
DOI:https://doi.org/10.1038/s41586-026-10228-w
Abstract
G-protein-coupled receptor (GPCR) signalling occurs through heterotrimeric G proteins, whose selective activation leads to distinct cellular outcomes1. Although more than 200 GPCR–G protein complex structures have been determined2, these static snapshots provide limited insight into the dynamics of G-protein association and dissociation. Here we present cryo-electron microscopy structures of human neurotensin receptor type 1 (NTSR1) with minimally modified Go and Gq, showing how the receptor’s intracellular surface dynamically rearranges to accommodate each G-protein subtype. Furthermore, time-resolved cryo-electron microscopy analyses of NTSR1–Gi visualized G-protein dissociation processes on GDP/GTP binding. Characterization of more than 20 intermediates, complemented by mutational and computational analyses, identifies four key mechanistic features. First, GDP/GTP induces Gi release from both canonical and non-canonical active conformations with distinct kinetics. Second, NTSR1 uses common intracellular rearrangements to recognize different G-protein subtypes and to promote activation of a single subtype. Third, separation from Gβγ involves stepwise remodelling of the Gα switches I–III. Finally, Gi dissociates from the receptor through a pathway that is distinct from that of Gs, and the canonical and non-canonical NTSR1–Gi complexes further diverge in their dissociation trajectories. These findings provide a comprehensive framework for understanding GPCR signalling dynamics and guiding signal-targeted therapeutic development.