2026-03-12 バージニア工科大学(Virginia Tech)
<関連情報>
- https://news.vt.edu/articles/2026/03/research-fralinbiomed-babychamp.html
- https://publications.aap.org/pediatricsopenscience/article/2/1/1/206520/Comparative-Efficacy-RCT-of-3-Intensive-Infant?autologincheck=redirected
片側性脳性麻痺に対する3つの集中的乳幼児療法の比較有効性RCT Comparative Efficacy RCT of 3 Intensive Infant/Toddler Therapies for Unilateral Cerebral Palsy
Stephanie C. DeLuca, PhD;Sharon Landesman Ramey, PhDCorresponding Author;Amy R. Darragh, PhD;Mark Conaway, PhD;Jill C. Heathcock, PhD;Warren Lo, MD;Andrew M. Gordon, PhD;Mary Rebekah Trucks, MS, OTR/L;Dory Wallace, MS, OTR/L;Thais Invencao Cabral, PhD
Pediatrics Open Science Published:February 20 2026
DOI:https://doi.org/10.1542/pedsos.2025-000676
OBJECTIVES
Unilateral cerebral palsy (UCP) can result in lifelong upper extremity (UE) neuromotor impairment. While both constraint-induced movement therapy (CIMT) and bimanual training have demonstrated efficacy for children with UCP, there was limited evidence to inform treatment decision-making in children aged between 6 and 24 months. Thus, we performed a comparative efficacy trial testing 3 high-dose therapist-delivered interventions, 2 CIMT interventions varied by constraint type to bimanual/no-constraint intervention for use in treating this age group of children with UCP.
PATIENTS AND METHODS
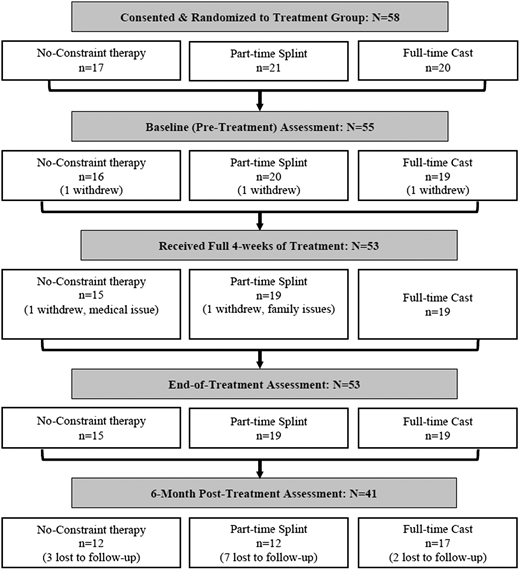
Fifty-eight infants/toddlers with UCP diagnosis, aged 6 to 24 months, were enrolled and randomized. Exclusion criteria were uncontrolled seizures, fragile health, prior CIMT/bimanual therapy, and recent botulinum toxin. Participants were randomly assigned (1:1:1) to 1 of 3 treatments all delivered 3 hours/d and 5 days/wk for 4 weeks: CIMT/full-time cast, CIMT/part-time splint, or bimanual/no constraint. Anonymized assessments at baseline, end of treatment (EoT), and 6 months posttreatment included the Mini–Assisting Hand Assessment (AHA) for bimanual abilities and the Bayley Scales of Infant and Toddler Development, Third Edition (Bayley-III) Fine-Motor (FM) subscale on each UE for FM abilities.
RESULTS
Fifty-three infant/toddlers completed treatment and EoT assessment (mean age, 17.2 months), and 41 completed 6-month assessment. All groups had gains from intervention: Mini-AHA scores (P < .003) and Bayley-III FM/paretic side (P < .002). Bayley-III FM/nonparetic side also improved across groups (P < .001). The CIMT/full-time cast showed larger gains on Bayley-III FM/nonparetic side when compared with bimanual/no constraint (difference, 5.9; 95% CI, 1.2-10.5; P = .015).
CONCLUSION
The trial confirms comparable benefits from therapist-delivered CIMT and bimanual/no-constraint interventions for infants/toddlers with UCP aged between 6 and 24 months.


