2026-03-18 カロリンスカ研究所(KI)
<関連情報>
- https://news.ki.se/immune-cells-are-programmed-within-the-body-to-fight-cancer
- https://www.nature.com/articles/s41586-026-10235-x
生体内における部位特異的な遺伝子操作によるT細胞の再プログラム化 In vivo site-specific engineering to reprogram T cells
William A. Nyberg,Pierre-Louis Bernard,Wayne Ngo,Charlotte H. Wang,Jonathan Ark,Allison Rothrock,Gina M. Borgo,Gabriella R. Kimmerly,Jae Hyung Jung,Vincent Allain,Jennifer R. Hamilton,Alisha Baldwin,Robert Stickels,Sarah Wyman,Safwaan H. Khan,Shanshan Lang,Donna Marsh,Niran Almudhfar,Catherine Novick,Yasaman Mortazavi,Shimin Zhang,Mahmoud M. AbdElwakil,Luis R. Sandoval,Sidney Hwang,… Justin Eyquem
Nature Published:18 March 2026
DOI:https://doi.org/10.1038/s41586-026-10235-x
Abstract
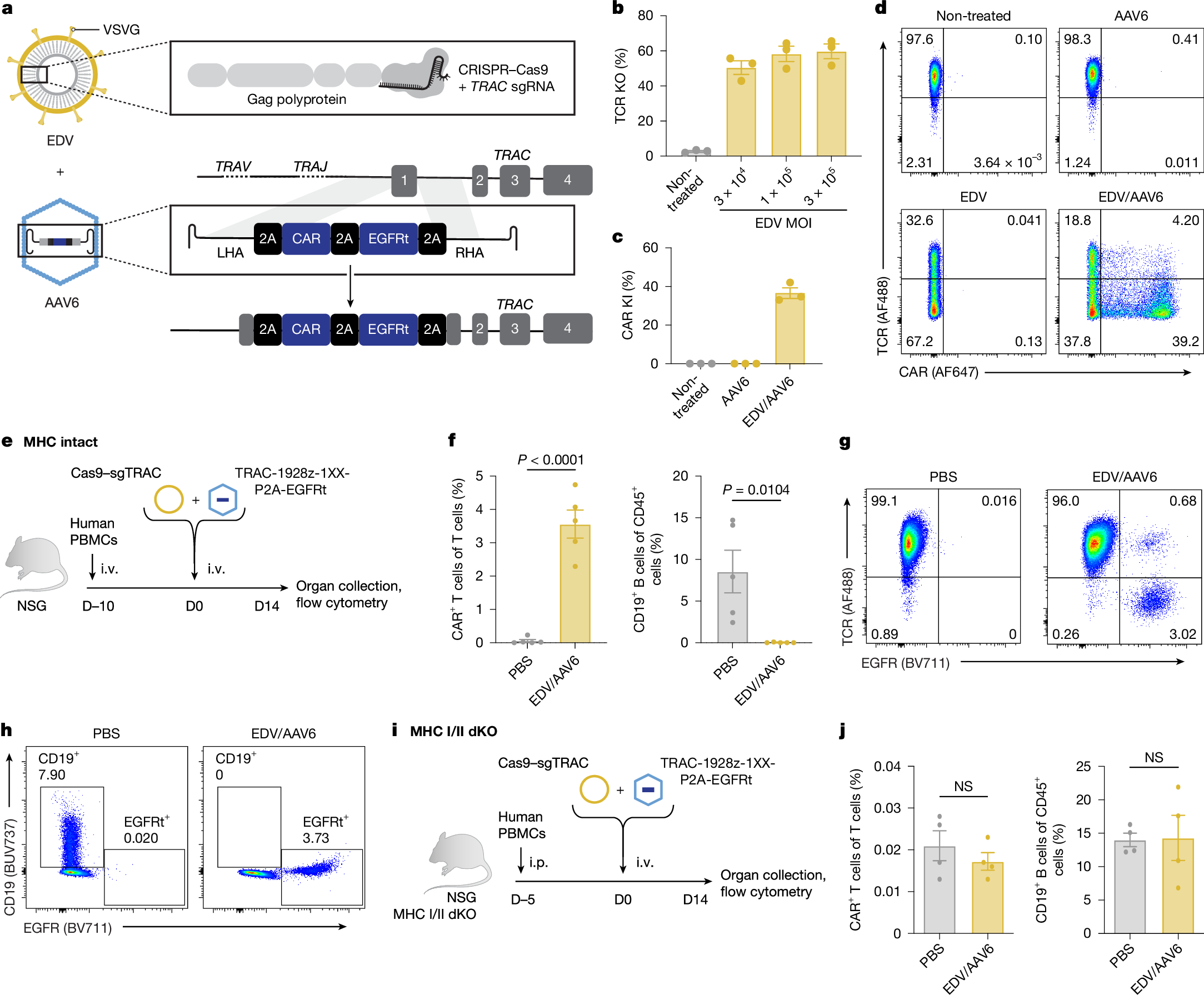
Engineered T cells, reprogrammed to express chimeric antigen receptors (CAR) or T cell receptors (TCR), have transformed cancer treatment and are being explored as therapeutics for autoimmune and infectious diseases. Enhancing T cell function through genome editing, either by disrupting endogenous genes or precisely inserting DNA payloads, has shown considerable promise1. However, the ex vivo manufacturing process is lengthy and costly, limiting accessibility of these therapies. In vivo generation of CAR T cells could overcome these barriers, but current methods rely either on transient expression with limited durability, or on random integration of DNA payloads that lack specificity. Here we demonstrate that stable and cell-specific transgene expression can be achieved through in vivo site-specific integration of large DNA payloads. We developed a two-vector system to deliver CRISPR–Cas9 ribonucleoproteins and a DNA donor template, using enveloped delivery vehicles and adeno-associated viruses, respectively. We optimized both vectors for T cell-specific delivery and gene-targeting efficiency. By integrating a CAR transgene into a T cell-specific locus, we generate therapeutic levels of CAR T cells in vivo in humanized mouse models of B cell aplasia, and haematological and solid malignancies. These findings offer a pathway to more efficient, precise and widely accessible T cell therapies.