2026-03-23 オックスフォード大学
<関連情報>
- https://www.ox.ac.uk/news/2026-03-23-blood-test-may-improve-survival-childhood-cancer-africa
- https://www.nature.com/articles/s41591-026-04291-z
流行地域におけるEBV陽性バーキットリンパ腫の診断のための液体生検 Liquid biopsy for the diagnosis of EBV-positive Burkitt’s lymphoma in endemic areas
Clara Chamba,Heavenlight Christopher,Emmanuel Josephat,Julius Sseruyange,Alisen Ayitewala,Kieran Howard,Helene Dreau,Adam Burns,Ismail Legason,Isaac Otim,Priscus Mapendo,Leah Mnango,Advera Ngaiza,Alex Mremi,Edrick Elias,Carol Achola,William Mawalla,Rehema Shungu,Eli Mkwizu,Lulu Chirande,Hadija Mwamtemi,Salama Mahawi,Godlove Sandi,Heronima J. Kashaigili,… Anna Schuh
Nature Medicine Published:19 March 2026
DOI:https://doi.org/10.1038/s41591-026-04291-z
Abstract
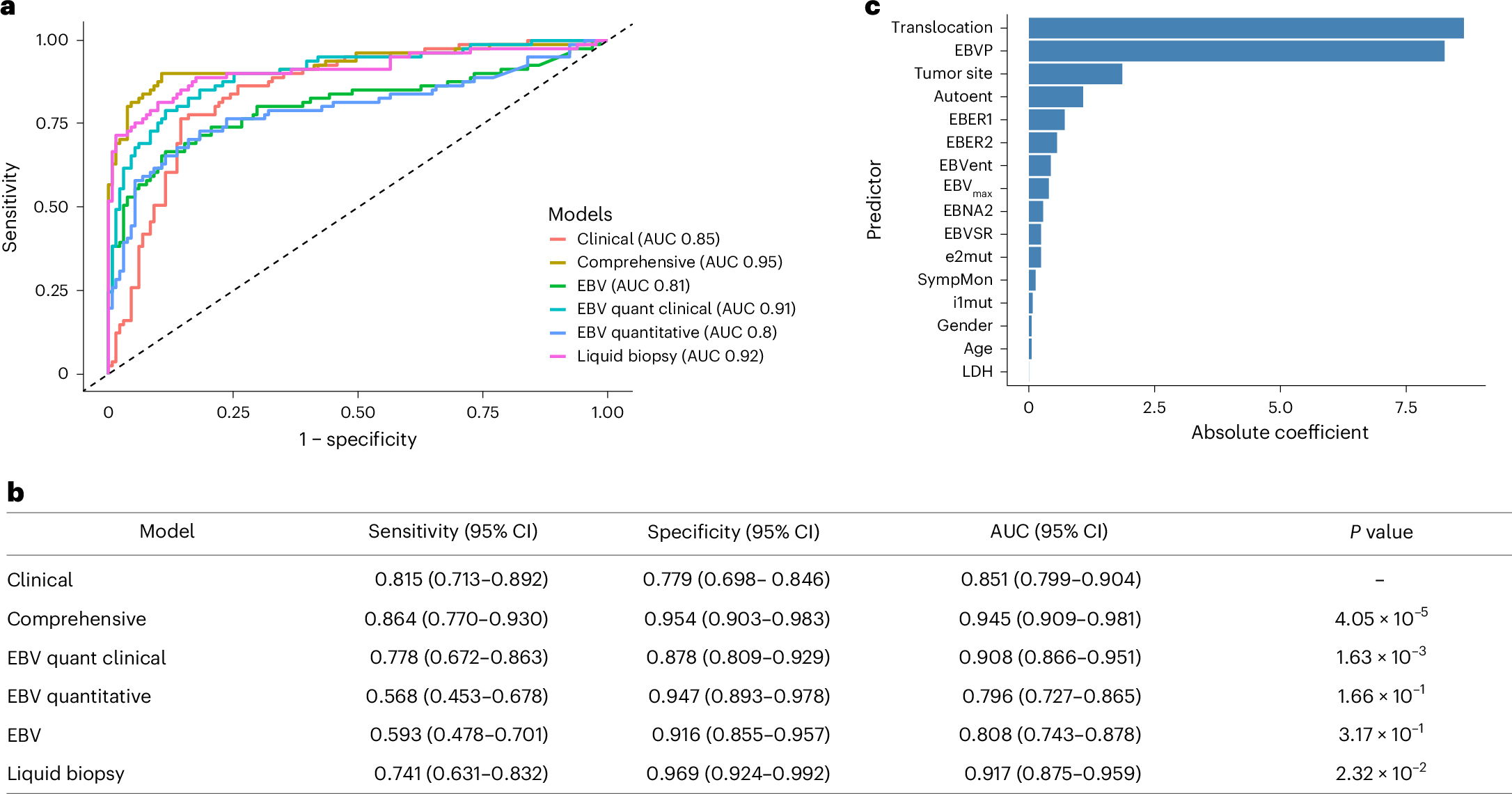
Burkitt’s lymphoma (BL) is common in sub-Saharan Africa, yet diagnosis is often delayed due to limited pathology capacity. Here we evaluated blood-based liquid biopsies from 377 children and young adults with clinically suspected lymphoma at four hospitals in Tanzania and Uganda, assessing diagnostic accuracy and turnaround time (TAT). After extensive pathology capacity building, a gold-standard diagnosis was established using tissue morphology, a limited validated immunohistochemistry panel and independent dual histopathologist review. Using clinical features and circulating tumor DNA markers (MYC mutations, MYC–immunoglobulin translocations and Epstein–Barr virus fragmentomics), we trained six penalized logistic regression models with tenfold crossvalidation (n = 212). The best-performing model was externally validated in a prospective real-world cohort (n = 56). Diagnostic accuracy, yield and TAT were compared head to head between liquid biopsy and the gold standard in 58 participants. The comprehensive model achieved the highest performance (area under the curve (AUC) 0.95, 95% confidence interval (95% CI) 0.901–0.981, sensitivity 0.86, specificity 0.95), confirmed by external validation (AUC 0.98, 95% CI 0.942–1.000). Liquid biopsy was the only diagnostic result available at the multidisciplinary review in 42% of participants and reduced median diagnostic TAT from 46.8 d to 6.5 d (P = 4.42 × 10−10). These findings demonstrate that liquid biopsy enables fast, highly accurate molecular diagnosis of EBV+ BL and may substantially reduce treatment delays in resource-limited settings.


