2026-04-09 合肥物質科学研究院(HFIPS)
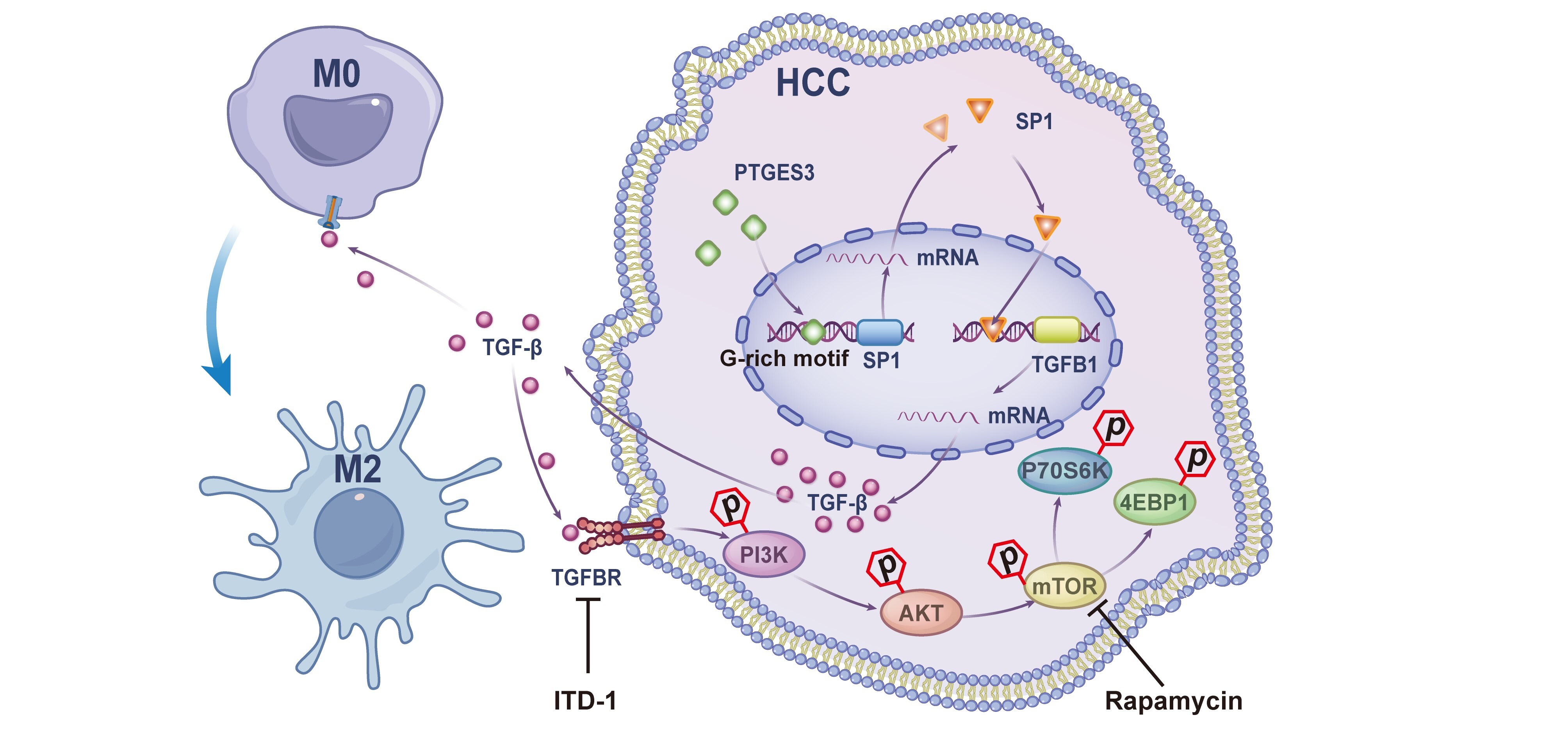
Schematic illustration showing the novel mechanism by which PTGES3 nuclear transcriptional regulation promotes hepatocellular carcinoma proliferation and immunosuppression. (Image by WANG Nianfei)
<関連情報>
- https://english.hf.cas.cn/nr/rn/202604/t20260409_1155581.html
- https://link.springer.com/article/10.1186/s43556-026-00431-6
核プロスタグランジンE合成酵素3は、SP1/TGF-β経路を介して免疫抑制性マクロファージ分極化により肝細胞癌の増殖を促進する Nuclear prostaglandin E synthase 3 promotes hepatocellular carcinoma growth with immunosuppressive macrophage polarization via the SP1/TGF-β axis
Nianfei Wang,Wei Chen,Shumin Shen,Jian Qi,Shanghu Wang,Rong Wang,Ming Li,Zixiang Chen,Jiangming Chen,Bo Hong & Hongzhi Wang
Molecular Biomedicine Published:25 March 2026
DOI:https://doi.org/10.1186/s43556-026-00431-6
Abstract
Hepatocellular carcinoma (HCC) is characterized by the synchronization of tumor cell proliferation and an immunosuppressive microenvironment. Decoupling these interconnected processes represents a major therapeutic challenge. Although Prostaglandin E Synthase 3 (PTGES3) functions canonically as a cytoplasmic Heat Shock Protein 90 (HSP90) co-chaperone, its non-canonical nuclear role in orchestrating tumor-immune crosstalk remains undefined. Here, we identify PTGES3 as a dual-function regulator coupling tumor intrinsic growth with extrinsic immune remodeling. We report that PTGES3 is upregulated in HCC and serves as an independent prognostic factor for poor survival. Using an immunocompetent, diethylnitrosamine (DEN)-induced HCC mouse model, we demonstrate that hepatocyte-specific Ptges3 silencing significantly suppresses tumorigenesis. Single-cell RNA sequencing (scRNA-seq) and histological analysis reveal that PTGES3 deficiency remodels the immune landscape, specifically by impairing tumor-associated macrophage (TAM) infiltration and M2 polarization. Mechanistically, we identified a specific G-rich motif on the Specificity Protein 1 (SP1) promoter bound by PTGES3 (confirmed via electrophoretic mobility shift assay [EMSA] and Cleavage Under Targets and Tagmentation [CUT&Tag]), which drives SP1-mediated Transforming Growth Factor-β (TGF-β) secretion. This axis appears to exert dual oncogenic effects: triggering paracrine M2 macrophage polarization to foster immunosuppression, whilst simultaneously fueling an autocrine TGF-β/TGFBR/PI3K/AKT/mTOR signaling loop to sustain tumor proliferation. Our findings define a non-canonical nuclear function for PTGES3, identifying it as a critical molecular switch that couples tumor aggressiveness with microenvironmental remodeling, thus presenting a promising therapeutic target for HCC.


