2022-08-15 フランクフルト大学
この研究で明らかになったように、シャペロンはプロセスの中心的な構成要素であり、厳格な選別を行うことによって、道路標識に長期的な安定性を与えているのである。大量の利用可能な材料の中から短命のウイルス断片を拒絶することで、MHC Iと複合体化した最良で最も安定したペプチドエピトープを装填したMHC I分子のみがペプチド装填複合体から放出されることを保証している。タパシンは、最適でないペプチドエピトープを最適なエピトープに交換するのを促進する触媒のような役割を果たします。
<関連情報>
- https://aktuelles.uni-frankfurt.de/englisch/road-signs-for-immune-defence-cells-study-generates-insights-into-inner-workings-of-adaptive-immune-response/
- https://www.nature.com/articles/s41467-022-32384-z
ペプチドローディング複合体におけるMHC I 品質管理の分子基盤 Molecular basis of MHC I quality control in the peptide loading complex
Alexander Domnick,Christian Winter,Lukas Sušac,Leon Hennecke,Mario Hensen,Nicole Zitzmann,Simon Trowitzsch,Christoph Thomas & Robert Tampé
Nature Communications Published:10 August 2022
DOI:https://doi.org/10.1038/s41467-022-32384-z
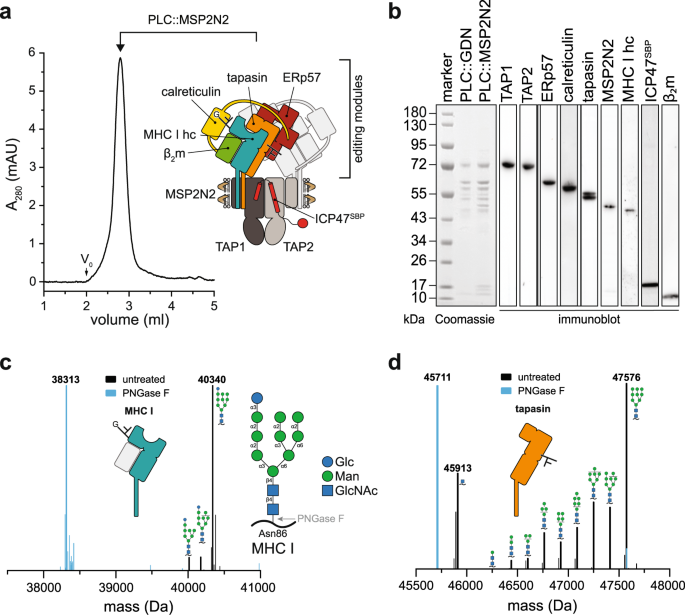
Abstract
Major histocompatibility complex class I (MHC I) molecules are central to adaptive immunity. Their assembly, epitope selection, and antigen presentation are controlled by the MHC I glycan through a sophisticated network of chaperones and modifying enzymes. However, the mechanistic integration of the corresponding processes remains poorly understood. Here, we determine the multi-chaperone-client interaction network of the peptide loading complex (PLC) and report the PLC editing module structure by cryogenic electron microscopy at 3.7 Å resolution. Combined with epitope-proofreading studies of the PLC in near-native lipid environment, these data show that peptide-receptive MHC I molecules are stabilized by multivalent chaperone interactions including the calreticulin-engulfed mono-glucosylated MHC I glycan, which only becomes accessible for processing by α-glucosidase II upon loading of optimal epitopes. Our work reveals allosteric coupling between peptide-MHC I assembly and glycan processing. This inter-process communication defines the onset of an adaptive immune response and provides a prototypical example of the tightly coordinated events in endoplasmic reticulum quality control.


