2024-03-01 パシフィック・ノースウェスト国立研究所(PNNL)
<関連情報>
- https://www.pnnl.gov/news-media/pnnl-scientists-help-pinpoint-molecular-switch-active-type-1-diabetes
- https://www.nature.com/articles/s41556-024-01368-0
β細胞におけるm6Aメチル化酵素METTL3のレドックス制御が1型糖尿病の自然免疫応答を制御する Redox regulation of m6A methyltransferase METTL3 in β-cells controls the innate immune response in type 1 diabetes
Dario F. De Jesus,Zijie Zhang,Natalie K. Brown,Xiaolu Li,Ling Xiao,Jiang Hu,Matthew J. Gaffrey,Garrett Fogarty,Sevim Kahraman,Jiangbo Wei,Giorgio Basile,Tariq M. Rana,Clayton Mathews,Alvin C. Powers,Audrey V. Parent,Mark A. Atkinson,Sirano Dhe-Paganon,Decio L. Eizirik,Wei-Jun Qian,Chuan He & Rohit N. Kulkarni
Nature Cell Biology Published:26 February 2024
DOI:https://doi.org/10.1038/s41556-024-01368-0
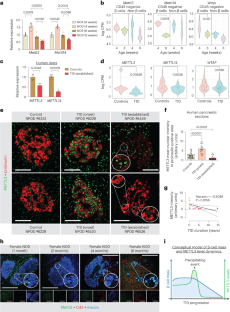
Abstract
Type 1 diabetes (T1D) is characterized by the destruction of pancreatic β-cells. Several observations have renewed the interest in β-cell RNA sensors and editors. Here, we report that N6-methyladenosine (m6A) is an adaptive β-cell safeguard mechanism that controls the amplitude and duration of the antiviral innate immune response at T1D onset. m6A writer methyltransferase 3 (METTL3) levels increase drastically in β-cells at T1D onset but rapidly decline with disease progression. m6A sequencing revealed the m6A hypermethylation of several key innate immune mediators, including OAS1, OAS2, OAS3 and ADAR1 in human islets and EndoC-βH1 cells at T1D onset. METTL3 silencing enhanced 2′-5′-oligoadenylate synthetase levels by increasing its mRNA stability. Consistently, in vivo gene therapy to prolong Mettl3 overexpression specifically in β-cells delayed diabetes progression in the non-obese diabetic mouse model of T1D. Mechanistically, the accumulation of reactive oxygen species blocked upregulation of METTL3 in response to cytokines, while physiological levels of nitric oxide enhanced METTL3 levels and activity. Furthermore, we report that the cysteines in position C276 and C326 in the zinc finger domains of the METTL3 protein are sensitive to S-nitrosylation and are important to the METTL3-mediated regulation of oligoadenylate synthase mRNA stability in human β-cells. Collectively, we report that m6A regulates the innate immune response at the β-cell level during the onset of T1D in humans.
