2026-02-27 京都大学
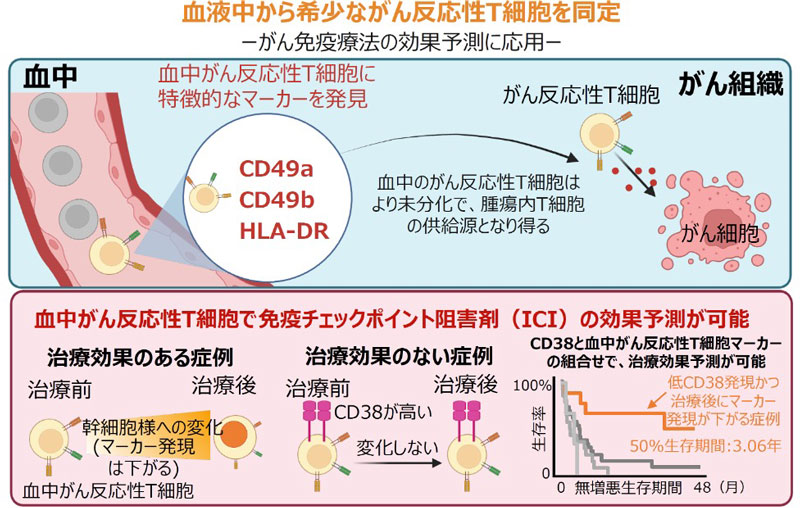
(上図)血液中にわずかに存在するがん反応性T細胞は特徴的な表面分子(CD49a、CD49b、HLA-DR)を発現し、がん組織内T細胞に比べ、疲弊が少なく未分化である。これらの特徴から供給源である可能性が示唆される。(下図)血中のがん反応性T細胞は、免疫チェックポイント阻害剤の治療効果の有無によって性質が異なり、効果予測マーカーとして応用可能である。(BioRenderで作成。作者:谷口智憲、伊藤克弘。)
<関連情報>
- https://www.kyoto-u.ac.jp/ja/research-news/2026-02-27-0
- https://www.kyoto-u.ac.jp/sites/default/files/2026-02/web_2602_Yaguchi-667b2934391f5a2b6e025dee2b345383.pdf
- https://www.nature.com/articles/s41467-026-69680-x
循環腫瘍反応性T細胞の表現型は非小細胞肺癌における免疫チェックポイント阻害剤の反応を予測する Phenotype of circulating tumor-reactive T cells predicts immune checkpoint inhibitor response in non-small cell lung cancer
Katsuhiro Ito,Kei Iida,Tomoko Hirano,Merrin Man Long Leong,Kenji Morii,Toshi Menju,Hiroshi Date,Hiroaki Ozasa,Hironori Yoshida,Toyohiro Hirai,Shusuke Kawashima,Kazuhiro Aoyama,Yuka Saeki,Takashi Inozume,Takashi Kobayashi,Kenji Chamoto & Tomonori Yaguchi
Nature Communications Published:17 February 2026
DOI:https://doi.org/10.1038/s41467-026-69680-x
We are providing an unedited version of this manuscript to give early access to its findings. Before final publication, the manuscript will undergo further editing. Please note there may be errors present which affect the content, and all legal disclaimers apply.
Abstract
Peripheral blood (PB) is a source of tumor-infiltrating tumor-reactive T cells (TR-T). Circulating TR-Ts (cTR-T) in PB are expected to contribute to the efficacy of immune checkpoint inhibitors (ICIs), but their phenotype remains poorly understood. Here we analyse paired tumor-infiltrating and peripheral CD8+ T cells from patients with non-small cell lung carcinoma (NSCLC), using single-cell RNA and T cell receptor (TCR) sequencing. Tumor-infiltrating TR-Ts are defined based on the reported TR-T-associated gene signatures. Using their TCR sequence as a barcode, we identify cTR-Ts and their specific surface markers, including CD49a, CD49b, and HLA-DR. Trajectory analysis assigns a progenitor-like phenotype to cTR-Ts, suggesting a potential developmental relationship with tumor-infiltrating TR-Ts. By single-cell transcriptomic and flow cytometric analysis on an ICI-treated cohort we show that pre-treatment cTR-Ts in responders are characterized by a relatively low expression of exhaustion-related CD38. Following the first dose, cTR-Ts of responders transit towards a TCF7+ stem-like phenotype. Additionally, we validate cTR-T’s phenotypic changes following PD-1 blockade therapy in mouse tumor models with artificial antigen. These findings suggest that the phenotypic state and transition of cTR-Ts may reflect their functional potential after tumor infiltration and are associated with therapeutic outcomes of ICIs.