2026-02-27 九州大学
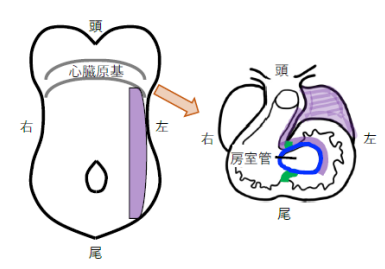
図1 受精 8 日目のマウス胚(左図)と受精 10 日目のマウス心臓(右図)の模式図受精 8 日胚は腹側から、受精 10 日心臓は前頭断面を腹側から眺めている。心臓内の緑色は心室間リングの一部を示す。紫色は Pitx2 を発現した左側側板中胚葉の細胞系譜であり、房室管(青色)と心室間リングの頭腹側で Pitx2 が発現する。
<関連情報>
- https://www.kyushu-u.ac.jp/ja/researches/view/1425
- https://www.kyushu-u.ac.jp/f/64968/26_0227_02.pdf
- https://insight.jci.org/articles/view/199072
心臓異位症における伝導系奇形はPitx2発現の調節不全に起因する Cardiac conduction system malformations in heterotaxy result from dysregulated Pitx2 expression
Kunihiko Joo, Ryohei Matsuoka, Keiko Kitajima, Kenta Yashiro, Akira Shiose, Ryuji Tominaga, Michael M. Shen, Shinya Oki, and Chikara Meno
JCI Insight Published: February 24, 2026
DOI:https://doi.org/10.1172/jci.insight.199072
Abstract
The cardiac conduction system (CCS) develops asymmetrically along the body axes. In heterotaxy syndrome—resulting from aberrant left–right (L–R) axis formation—atrial and atrioventricular conduction defects can cause life-threatening arrhythmias. However, the developmental mechanisms regulating the atrioventricular conduction system (AVCS) disposition and integrity remain unclear. To investigate the etiology of AVCS malformations in laterality defects, we analyzed CCS development and function in mouse mutants for Cryptic and Lefty1, which are key regulators of Pitx2 in the L–R axis formation. Cryptic–/– embryos exhibited bilateral sinoatrial (SA) nodes and an ectopic anterior AV node and bundle accompanied by reduced Pitx2 expression. In contrast, Lefty1–/– embryos showed a hypoplastic SA node and AV node–bundle dissociation with ectopic Pitx2 expression. Single-cell transcriptomic analysis of Pitx2–/– hearts revealed expansion of AV node and bundle populations, consistent with a repressive role of Pitx2 in AVCS specification. Genetic lineage tracing indicated that Pitx2-expressing cells from the left lateral plate mesoderm populate cranioventral cardiac regions, where AVCS development is suppressed. Together, these findings clarify how global L–R axis information is locally integrated to shape AVCS disposition and integrity, providing a mechanistic model for AVCS abnormalities in laterality-associated congenital heart disease.