2026-03-04 北海道大学,東北大学
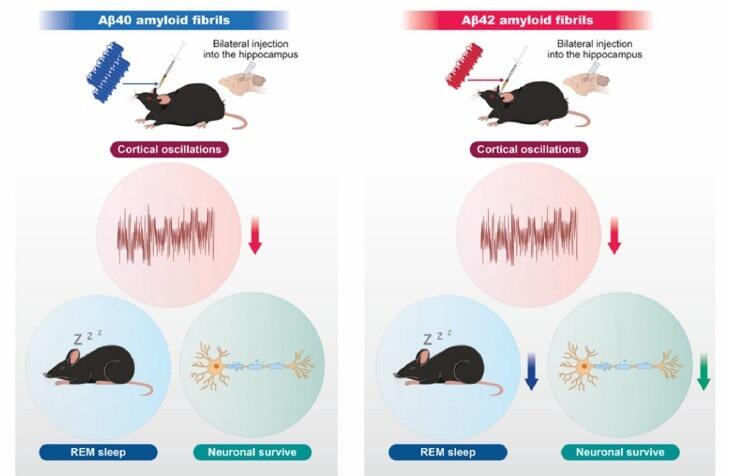
線維化Aβ40または線維化Aβ42をマウス海馬へ両側投与し、睡眠・覚醒状態及び皮質脳波活動(皮質振動)を解析した。両群で皮質脳波の変化が認められたが、線維化Aβ42ではレム睡眠の減少に加え、神経細胞脱落が示唆された。
<関連情報>
- https://www.hokudai.ac.jp/news/2026/03/a40a42.html
- https://www.hokudai.ac.jp/news/pdf/260304_pr.pdf
- https://www.sciencedirect.com/science/article/pii/S0301462226000323
アルツハイマー病関連アミロイド線維は、その種類によってマウスの睡眠と皮質脳波活動を異なる形で変化させる Amyloid fibrils in Alzheimer’s disease differently modulate sleep and cortical oscillations in mice depending on the type of amyloid
Tomomi Sanagi, Masaki Okumura, Yuxi Lin, Shingo Kanemura, Eunyoung Moon, Yunseok Heo, Keiko Takahara, Young-Ho Lee, Tomomi Tsunematsu
Biophysical Chemistry Available online: 25 February 2026
DOI:https://doi.org/10.1016/j.bpc.2026.107599
Highlights
- Aβ42 monomers show faster amyloid formation than Aβ40 monomers.
- Aβ42 forms structurally and morphologically distinct amyloids compared to Aβ40.
- Aβ42 fibrils markedly disrupt REM sleep and alter cortical oscillations.
- Aβ40 fibrils mainly affect cortical activity with minimal neuronal loss.
- Distinct properties of amyloids contribute to differences in sleep disruption.
Abstract
Alzheimer’s disease (AD) is characterized by aggregation and deposition of the amyloid-beta (Aβ) protein in patients’ brains, with aging contributing through oxidative stress and neuroinflammation. Sleep disturbances are common in patients with AD and exacerbate cognitive impairment. However, it remains unclear how aggregation of specific Aβ species in distinct brain regions contributes to sleep dysfunction. To address this, we characterized amyloid fibrils formed by Aβ1–40 (Aβ40) and Aβ1–42 (Aβ42) by assessing their structural and surface properties using fluorescence, CD, and NMR spectroscopy. Their morphology was also visualized using TEM and AFM. These analyses revealed that Aβ42 aggregates faster than Aβ40 and forms amyloid fibrils with distinct structural, surface, and morphological properties. To investigate their effects in vivo, we bilaterally injected Aβ40 and Aβ42 fibrils into the hippocampus of wild-type mice and recorded the electroencephalogram and electromyogram under freely moving conditions. Aβ42 amyloid fibrils significantly disrupted sleep architecture, particularly REM sleep, and altered oscillations, accompanied by neuronal loss. In contrast, Aβ40 amyloid fibrils mainly affected cortical activity with minimal neuronal loss and caused comparatively modest changes in sleep. These findings demonstrate that a single administration of Aβ amyloid fibril is sufficient to alter sleep/wakefulness state and cortical oscillations. The observed effects differ depending on the type of Aβ administered, suggesting that the physicochemical properties of the fibrils are closely linked to their capacity to induce sleep impairment. These findings shed light on AD-associated sleep disorders, which are differentially affected by the distinct properties of Aβ40 and Aβ42 aggregates.