2026-04-01 東京大学
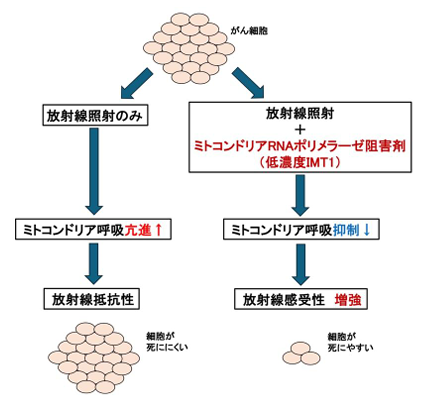
ミトコンドリアRNAポリメラーゼ(POLRMT)阻害による放射線増感の概念図
<関連情報>
- https://www.u-tokyo.ac.jp/content/400283179.pdf
- https://academic.oup.com/jrr/advance-article/doi/10.1093/jrr/rrag021/8553207
ミトコンドリアRNAポリメラーゼの阻害は、ミトコンドリア呼吸を阻害することにより、がん細胞の放射線感受性を高める Inhibition of mitochondrial RNA polymerase sensitizes cancer cells to radiation by inhibiting mitochondrial respiration
Sachiko Tsunoda ,Yukina Osawa ,Noriko Hosoya
Journal of Radiation Research Published:01 April 2026
DOI:https://doi.org/10.1093/jrr/rrag021
Abstract
Radiotherapy is a cornerstone of cancer treatment, but its efficacy is limited by tumor radioresistance and toxicity to normal cells. Thus, radiosensitizing agents that selectively target cancer-specific pathways are needed. Mitochondria contain their own deoxyribonucleic acid (DNA) that encodes proteins essential for oxidative phosphorylation (OXPHOS), the primary energy source for cell growth and survival. Recently, IMT1, a specific inhibitor of mitochondrial transcription targeting mitochondrial RNA polymerase (POLRMT), was developed and shown to suppress tumor growth in several cancers overexpressing POLRMT. However, the effect of combining IMT1 with radiation remains uncharacterized. Here, we show that IMT1 enhances radiosensitivity in cancer cells by inhibiting mitochondrial respiration. We inhibited POLRMT by administrating sublethal dose of IMT1 in OXPHOS-dependent cancer cell lines HeLa, A549, MDA-MB-468, HCT116, A431 and AN3CA, and observed increased radiosensitivity. While radiation alone upregulated mitochondrial respiration, IMT1 abolished this capacity when combined with radiation, showing very low oxygen consumption rates in all respiratory states. IMT1 enhanced radiation-induced apoptosis, but did not affect DNA damage repair and cell cycle regulation. Supplementation with galactose rescued hyper-radiosensitivity induced by IMT1. These findings support the mechanistic link between impaired mitochondrial respiration and radiosensitization induced by POLRMT inhibition. The radiosensitizing effect of IMT1 was not observed in normal cell lines RPE1 and HME1 and the glycolysis-dominant cancer cell line HT1080, suggesting that OXPHOS-dominant cancers would profit most from POLRMT inhibition. Thus, this study presents a novel therapeutic strategy that may improve the efficacy of radiotherapy in OXPHOS-dependent cancer cells while minimizing damage to normal cells.