2026-04-02 国立循環器病研究センター
<関連情報>
- https://www.ncvc.go.jp/pr/release/pr_51743/
- https://www.neurology.org/doi/10.1212/WNL.0000000000214745
抗血栓療法を受けている患者における、基底核周囲血管腔が虚血性および出血性リスクに及ぼす影響 Impact of Basal Ganglia Perivascular Spaces on Ischemic and Hemorrhagic Risks in Patients Taking Antithrombotic Therapies
Soya Iwamoto, Kaori Miwa, Masatoshi Koga, Sohei Yoshimura, Kanta Tanaka, Yusuke Yakushiji, Makoto Sasaki, …for the BAT2 Investigators
Neurology Published:March 20, 2026
DOI:https://doi.org/10.1212/WNL.0000000000214745
Abstract
Background and Objectives
Enlarged perivascular spaces (ePVSs) are considered to increase the risk of stroke. However, data regarding the benefits and risks of antithrombotic therapy in patients with ePVSs are currently limited. This study assessed the association between ePVSs and the risks of hemorrhagic or ischemic events in patients taking antithrombotic agents.
Methods
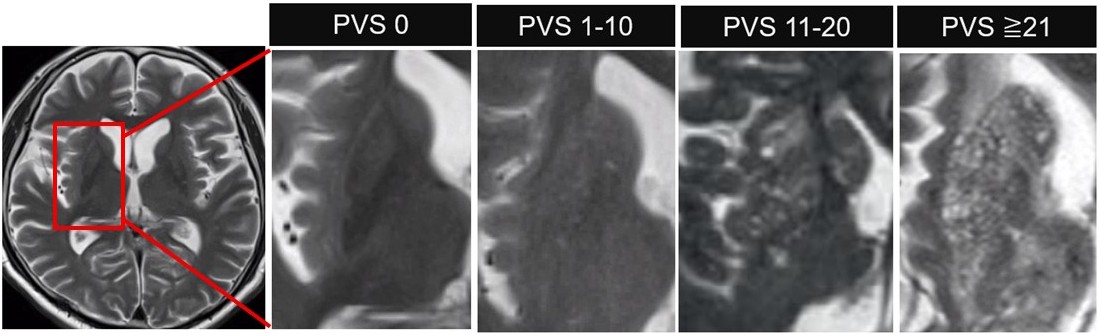
This prospective, multicenter observational study enrolled patients with cerebrovascular or cardiovascular diseases who had newly started or were continuing to take oral antithrombotic agents at 52 hospitals across Japan between 2016 and 2019. Baseline multimodal MRI was performed for all of the study participants. The images were then centrally evaluated for cerebral small vessel disease (SVD), including white matter hyperintensities, cerebral microbleeds, lacunes, and basal ganglia–enlarged perivascular spaces (BGPVSs). BGPVSs were categorized as 0, 1–10, 11–20, or ≥21. Outcomes included major bleeding, intracranial hemorrhage, ischemic events, ischemic stroke, and mortality. Multivariable Cox proportional hazards models were used for the BGPVSs (in categorical and ordinal forms), adjusted for patient demographics, vascular risk factors, and other SVD markers.
Results
Of the total 5,065 patients (1,663 women; median age 74 [interquartile range 67–81] years), antiplatelets and anticoagulants were administered at the baseline to 3,820 (75.4%) and 1,502 (29.7%) patients, respectively. Their BGPVS distributions were as follows: 0, 475 (9.4%); 1–10, 2,615 (51.6%); 11–20, 1,267 (25.0%); and 21+, 708 patients (14.0%). Over a median follow-up period of 2.0 (interquartile range 1.8–2.0) years, we noted 266 ischemic events, 188 ischemic strokes, 92 major bleeding events, 54 intracranial hemorrhages, and 198 deaths. Higher BGPVS was associated with an increased risk of major bleeding (BGPVS ≥21: adjusted hazard ratio [aHR] 4.04, 95% CI 1.17–13.92; per-unit increase: aHR 1.38, 95% CI 1.07–1.77) and ischemic stroke (BGPVS ≥21: aHR 2.58, 95% CI 1.21–5.50; per-unit increase: aHR 1.28, 95% CI 1.07–1.53) but was not significantly associated with higher risks of ischemic events, intracranial hemorrhage, or mortality.
Discussion
Higher BGPVS burdens are associated with higher risks of ischemic stroke and major bleeding in patients receiving antithrombotic therapy. BGPVSs may serve as useful imaging biomarkers for vascular risk assessment and personalized therapy.