2026-04-07 国立精神・神経医療研究センター
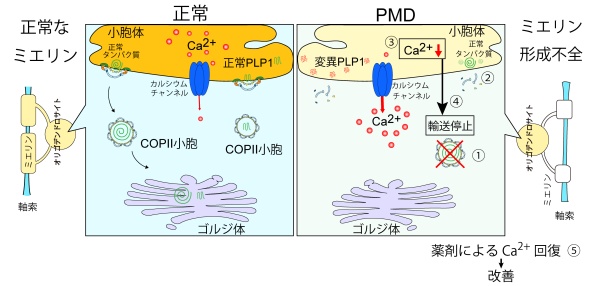
概要図:本研究で明らかになった新規病態モデル
<関連情報>
- https://www.ncnp.go.jp/topics/detail.php?@uid=gRLrknF5rEubBsPh
- https://www.sciencedirect.com/science/article/pii/S0969996126000732
変異型PLP1は、ペリツァウス・メルツバッハー病においてERカルシウム枯渇を介してCOPII小胞形成を阻害する Mutant PLP1 impairs COPII vesicle formation via ER calcium depletion in Pelizaeus-Merzbacher disease
Heng Li, Reiko Mishima, Yu-ichi Goto, Ken Inoue
Neurobiology of Disease Available online: 24 February 2026
DOI:https://doi.org/10.1016/j.nbd.2026.107329
Highlights
- Mutant PLP1 depletes ER Ca2+ in oligodendrocytes.
- ER Ca2+ depletion disrupts ALG-2/Sec31A/AnxA11 complex.
- COPII formation is impaired, causing global ER-Golgi trafficking failure.
- ER Ca2+ depletion–induced ER-Golgi trafficking failure underlies PMD pathology.
Abstract
Pelizaeus-Merzbacher disease (PMD) is a devastating, X-linked hypomyelinating leukodystrophy caused by mutations in a myelin gene, PLP1. While overwhelming endoplasmic reticulum (ER) stress caused by the accumulation of mutant PLP1 is widely recognized, blockade of the apoptotic arm of the unfolded protein response (UPR) failed to rescue the phenotypes in murine disease models, suggesting the involvement of additional, critical cellular mechanisms in oligodendrocyte dysfunction. Herein, we identified ER Ca2+ depletion and disrupted ER-Golgi trafficking as key cellular pathologies in PMD. Mutant PLP1 impairs COPII vesicle formation by destabilizing its key components, including Sec31A at ER exit sites due to the Ca2+ transport dysregulation and deconstruction of the ALG-2/Sec31A/AnxA11 interaction. Pharmacological restoration of ER Ca2+ levels rescued COPII formation. These findings highlight how PLP1 mutations affect the intracellular trafficking of membrane and secretory proteins through the ER Ca2+ depletion, which may be associated with the clinical consequences of PMD and other inherited myelin disorders.