2026-04-10 星薬科大学
【研究概要図】
<関連情報>
- https://www.hoshi.ac.jp/news/news-study/44133/
- https://www.hoshi.ac.jp/wp/wp-content/uploads/2026/04/202604101400_narita.pdf
- https://www.sciencedirect.com/science/article/pii/S1043661826000289
感覚ニューロンを介した疼痛シグナル伝達は、神経ペプチド放出とκ-オピオイド拮抗作用を介して乳がんの進行を促進する Pain signaling via sensory neurons drives breast cancer progression through neuropeptide release and κ-opioid counter-regulation
Hitoshi Makabe, Michiko Narita, Yasuyuki Nagumo, Masanori Fujiwara, Yusuke Hamada, Jion Takise, Takumi Yoshizawa, Sakura Sano, Shin Iizuka, Eri Asaba, Yukari Suda, Tomohisa Mori, Tsuyoshi Saitoh, Hiroshi Nagase, Vivianne L. Tawfik, Shigehiro Yagishita, Akinobu Hamada, Kan Yonemori, Shin Takayama, Masayuki Yoshida…Minoru Narita
Pharmacological Research Available online: 28 January 2026
DOI:https://doi.org/10.1016/j.phrs.2026.108113
Highlights
- Elevated CGRP/SP levels link sensory neural signaling to reduced patient survival.
- Nociceptive neural activation accelerates breast cancer growth via CGRP/SP.
- Sensory neuron-derived CGRP/SP promotes EMT and chemoresistance in TNBC cells.
- In vivo blockade of CGRP/SP receptors suppresses tumor progression in PDX models.
- Dynorphin–KOR signaling attenuates breast cancer growth.
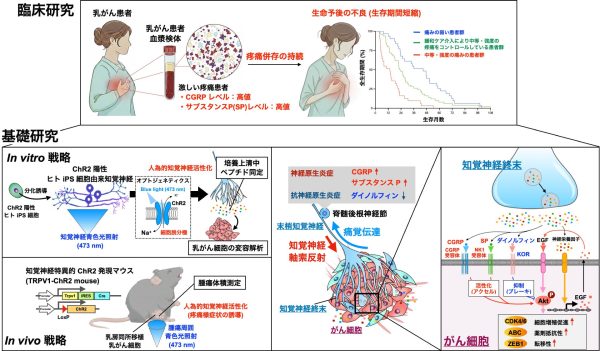
Abstract
Severe pain in patients with breast cancer is associated with poorer survival outcomes, yet the mechanisms linking nociceptive signaling to tumor progression remain unclear. In a clinical cohort, breast cancer patients whose moderate or severe pain improved under specialized palliative care exhibited significantly longer survival than those with persistent pain. Serum levels of calcitonin gene-related peptide (CGRP) and substance P (SP), neuropeptides that can be released from sensory neurons, were elevated in breast cancer patients with uncontrolled pain, correlating positively with pain intensity and negatively with overall survival. In vitro, supernatants from depolarized human iPSC-derived sensory neurons containing CGRP, SP, and the endogenous κ-opioid receptor (KOR) ligand dynorphin attenuated docetaxel efficacy, promoted epithelial-mesenchymal transition, and enhanced migration in human triple-negative breast cancer (TNBC) cells, accompanied by Gi protein-coupled activation of the PI3K-Akt signaling pathway. In vivo, optogenetic activation of sensory neurons significantly accelerated tumor growth following orthotopic transplantation of murine TNBC cells, whereas combined blockade of CGRP and SP receptors suppressed this effect. Similarly, in patient-derived xenograft models of TNBC, dual receptor blockade effectively abrogated tumor progression. Conversely, in vitro treatment with dynorphin enhanced the cytotoxic efficacy of docetaxel, accompanied by the inhibition of PI3K-Akt signaling, whereas in vivo administration of the peripherally restricted KOR agonist nalfurafine methiodide markedly suppressed tumor growth. These findings establish a bidirectional neural-tumor axis in breast cancer progression. Therapeutically, combined blockade of CGRP/SP signaling with peripheral KOR agonism may present a novel strategy to enhance chemotherapy efficacy in TNBC patients.