2026-04-13 慶應義塾大学,科学技術振興機構
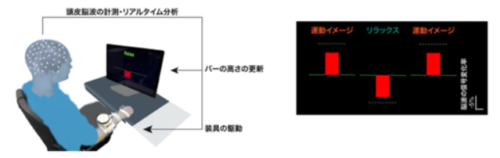
図1. ブレイン・コンピューター・インターフェースの操作訓練
<関連情報>
- https://www.keio.ac.jp/ja/press-release/20260413-press-01/
- https://www.keio.ac.jp/files/ae4736c53fae6b57ba0153aeb1f4d78adb80c9147f85800172c24b06cde3b656
- https://www.pnas.org/doi/10.1073/pnas.2525769123
ブレイン・コンピュータ・インターフェースを用いたニューロフィードバック訓練は、ヒトにおける皮質状態切り替えの汎化可能な制御を可能にする Brain–computer interface–based neurofeedback training enables transferable control of cortical state switching in humans
Seitaro Iwama, Atsuya Matsuoka, and Junichi Ushiba
Proceedings of the National Academy of Sciences Published:April 10, 2026
DOI:https://doi.org/10.1073/pnas.2525769123
Significance
Humans can flexibly control actions. Although closed-loop regulation of sensorimotor activity has been demonstrated since the 1960s, whether these volitional transitions can be rapidly controlled to improve behavior was unknown. Here, we demonstrate that humans can learn to self-regulate sensorimotor rhythms (SMR) using a brain–computer interface to master volitional switching between distinct cortical states defined by SMR amplitude. This learned control was associated with modulated neural dynamics in the sensorimotor network and characterized by rapid, large-scale shifts in periodic and aperiodic brain activity. Critically, the acquired skill generalized to overt motor tasks, significantly shortening reaction times for both muscle contraction and relaxation, suggesting that both reward-based and brain-state–contingent learning mechanisms contribute to intercontext generalization and enable mental practice gains.
Abstract
Behavioral flexibility relies on transient neural dynamics that govern cortical state transitions. However, whether humans can deliberately learn to control such state transitions and generalize trained neural dynamics beyond contexts remains unclear. Here, we demonstrate that operation of a brain–computer interface (BCI) which links time evolution of sensorimotor activity with real-time feedback enables volitional control over the targeted neural population. Compared with a double-blind sham control group, trained participants modulated sensorimotor oscillations in the absence of BCI. Data-driven latent-state analysis further revealed stronger interregional phase coupling and steeper broadband spectral slope in the medial frontal cortex during transitions. The training-induced reorganization of sensorimotor dynamics was found during movement execution and associated with performance improvement, indexed by reduced reaction times for both muscle contraction and relaxation. These findings provide evidence that learned control over cortical state transitions enhances behavioral flexibility beyond the training context.