2026-05-12 千葉大学医学部附属病院,量子科学技術研究機構
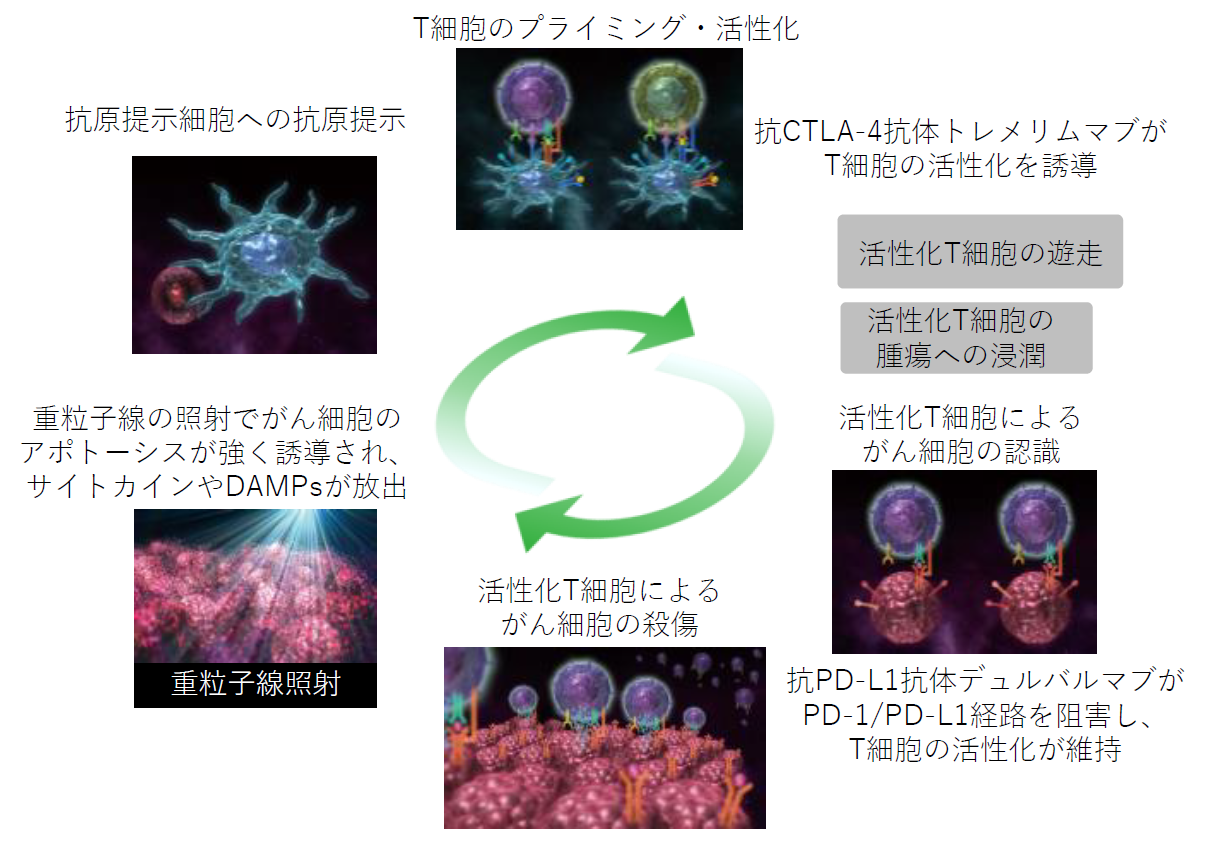
図: 免疫療法(抗PD-L1抗体+抗CTLA-4抗体)と重粒子線治療との併用ががん免疫サイクルに与える好循環(イメージ)
<関連情報>
- https://www.qst.go.jp/site/press/20260512.html
- https://www.jhep-reports.eu/article/S2589-5559(26)00036-4/fulltext
進行性肝細胞癌に対するMVI標的炭素イオン線治療と免疫療法の併用:第Ib相DEPARTURE試験 MVI-targeted carbon-ion radiotherapy combined with immunotherapy for advanced hepatocellular carcinoma: Phase Ib DEPARTURE trial JHEP Reports Published:February 5, 2026 DOI:https://doi.org/10.1016/j.jhepr.2026.101765
Highlights
- The trial evaluated the C-ion RT plus ICIs for HCC with macrovascular invasion.
- No DLTs were observed, and the combination showed a manageable safety profile.
- Many patients experienced transient high fevers after C-ion RT.
- The approach achieved safety and effective local control.
- The results highlight the need for further optimization of this combination strategy.
Abstract
Background & Aims
Advanced hepatocellular carcinoma (HCC) with macrovascular invasion (MVI) carries an extremely poor prognosis, necessitating novel therapeutic strategies. This phase Ib trial evaluated the safety and preliminary efficacy of combining carbon-ion radiotherapy (C-ion RT) with immune checkpoint inhibitors (ICIs) in patients with advanced HCC with MVI.
Methods
Fifteen patients with MVI-positive advanced HCC were enrolled (Cohort A: durvalumab monotherapy, n = 3; Cohort B: durvalumab plus tremelimumab, n = 12). C-ion RT (60 Gy, four fractions) was delivered to the MVI-containing primary tumor, while systemic therapy with durvalumab (+tremelimumab) was administered concurrently. The primary endpoints included dose-limiting toxicities and adverse events. Secondary endpoints included progression-free survival and overall survival.
Results
No dose-limiting toxicities were observed, and the combination exhibited a manageable safety profile. The most common adverse events were pyrexia, rash, and elevated lipase levels. Grade 3–4 adverse events occurred in 53.3%, including cytokine release syndrome and meningitis. Median progression-free survival and overall survival were 4.7 and 10.4 months, respectively. Although C-ion RT achieved effective local control of irradiated lesions, non-irradiated lesions showed limited systemic immune responses.
Conclusions
The combination of MVI-targeted C-ion RT and immune checkpoint inhibitors demonstrated safe and effective local tumor control in advanced HCC. This novel approach of selective irradiation to MVI-containing tumors, combined with systemic immunotherapy, warrants further investigation to optimize the synergistic effects and enhance systemic efficacy in this poor-prognosis group.
Impact and implications
Advanced hepatocellular carcinoma with macrovascular invasion (MVI) has a poor prognosis, highlighting the need for new therapeutic strategies. Our phase Ib study suggests that carbon-ion radiotherapy targeting MVI combined with immune checkpoint inhibitors is feasible and achieves sustained local tumor control. RNA-sequencing revealed that immune activation pathways were enriched in responders, while resistance was associated with mesenchymal and angiogenesis signatures. These results reinforce the potential of MVI-targeted irradiation combined with immune checkpoint inhibitors as a promising treatment strategy for these high-risk patients, warranting further investigation to improve systemic tumor control.
Clinical Trials Registration
jRCT2031210046.