2024-05-06 バーミンガム大学
<関連情報>
- https://www.birmingham.ac.uk/news/2024/red-light-therapy-for-repairing-spinal-cord-injury-passes-milestone
- https://aiche.onlinelibrary.wiley.com/doi/10.1002/btm2.10674
脊髄損傷後の神経再生と失われた機能の回復を促進する埋め込み型および経皮的光バイオモジュレーション Implantable and transcutaneous photobiomodulation promote neuroregeneration and recovery of lost function after spinal cord injury
Andrew R. Stevens, Mohammed Hadis, Alice Phillips, Abhinav Thareja, Michael Milward, Antonio Belli, William Palin, David J. Davies, Zubair Ahmed
Bioengineering & Translational Medicine Published: 25 April 2024
DOI:https://doi.org/10.1002/btm2.10674

Abstract
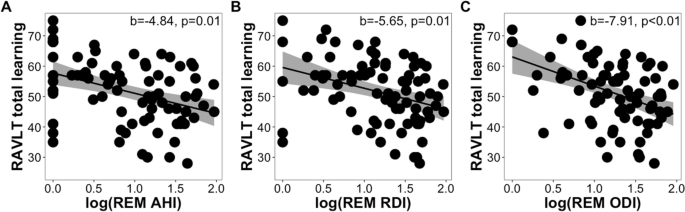
Spinal cord injury (SCI) is a cause of profound and irreversible damage, with no effective therapy to promote functional recovery. Photobiomodulation (PBM) may provide a viable therapeutic approach using red or near-infrared light to promote recovery after SCI by mitigating neuroinflammation and preventing neuronal apoptosis. Our current study aimed to optimize PBM dose regimens and develop and validate the efficacy of an invasive PBM delivery paradigm for SCI. Dose optimization studies were performed using a serum withdrawal model of injury in cultures of primary adult rat dorsal root ganglion neurons (DRGN). Implantable and transcutaneous PBM delivery protocols were developed and validated using cadaveric modeling. The efficacy of PBM in promoting recovery after SCI in vivo was studied in a dorsal column crush injury model of SCI in adult rats. Optimal neuroprotection in vitro was achieved between 4 and 22 mW/cm2. 11 mW/cm2 for 1 min per day (0.66 J/cm2) increased cell viability by 45% over 5 days (p <0.0001), increasing neurite outgrowth by 25% (p <0.01). A method for invasive application of PBM was developed using a diffusion-tipped optogenetics fiber optic. Delivery methods for PBM were developed and validated for both invasive (iPBM) and noninvasive (transcutaneous) (tcPBM) application. iPBM and tcPBM (24 mW/cm2 at spinal cord, 1 min per day (1.44 J/cm2) up to 7 days) increased activation of regeneration-associated protein at 3 days after SCI, increasing GAP43+ axons in DRGN from 18.0% (control) to 41.4% ± 10.5 (iPBM) and 45.8% ± 3.4 (tcPBM) (p <0.05). This corresponded to significant improvements at 6 weeks post-injury in functional locomotor and sensory function recovery (p <0.01), axonal regeneration (p <0.01), and reduced lesion size (p <0.01). Our results demonstrated that PBM achieved a significant therapeutic benefit after SCI, either using iPBM or tcPBM application and can potentially be developed for clinical use in SCI patients.
Translational Impact Statement
Photobiomodulation (PBM) may improve recovery after spinal cord injury (SCI) yet delivery of PBM to the injured spinal cord is limited by overlying tissue when applying PBM through the skin. We show the development and use of an implantable device for application of PBM which addresses this problem. Our results demonstrated that implantable PBM improved regeneration of neurons and these doses of PBM also improved functional recovery. This device has the potential to be developed to deliver PBM aimed at improving recovery in patients with SCI.