2025-01-30 スイス連邦工科大学ローザンヌ校 (EPFL)
<関連情報>
- https://actu.epfl.ch/news/designing-proteins-with-their-environment-in-mind/
- https://www.nature.com/articles/s41557-024-01719-2
溶媒を介したアロステリックネットワークによる高信号活性膜受容体の計算デザイン Computational design of highly signalling-active membrane receptors through solvent-mediated allosteric networks
K.-Y. M. Chen,J. K. Lai,L. S. P. Rudden,J. Wang,A. M. Russell,K. Conners,M. E. Rutter,B. Condon,F. Tung,L. Kodandapani,B. Chau,X. Zhao,J. Benach,K. Baker,E. J. Hembre & P. Barth
Nature Chemistry Published:23 January 2025
DOI:https://doi.org/10.1038/s41557-024-01719-2
Abstract
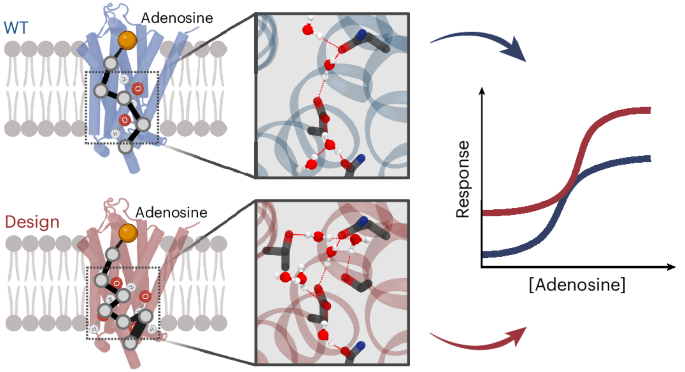
Protein catalysis and allostery require the atomic-level orchestration and motion of residues and ligand, solvent and protein effector molecules. However, the ability to design protein activity through precise protein–solvent cooperative interactions has not yet been demonstrated. Here we report the design of 14 membrane receptors that catalyse G protein nucleotide exchange through diverse engineered allosteric pathways mediated by cooperative networks of intraprotein, protein–ligand and –solvent molecule interactions. Consistent with predictions, the designed protein activities correlated well with the level of plasticity of the networks at flexible transmembrane helical interfaces. Several designs displayed considerably enhanced thermostability and activity compared with related natural receptors. The most stable and active variant crystallized in an unforeseen signalling-active conformation, in excellent agreement with the design models. The allosteric network topologies of the best designs bear limited similarity to those of natural receptors and reveal an allosteric interaction space larger than previously inferred from natural proteins. The approach should prove useful for engineering proteins with novel complex protein binding, catalytic and signalling activities.