2026-05-08 東北大学
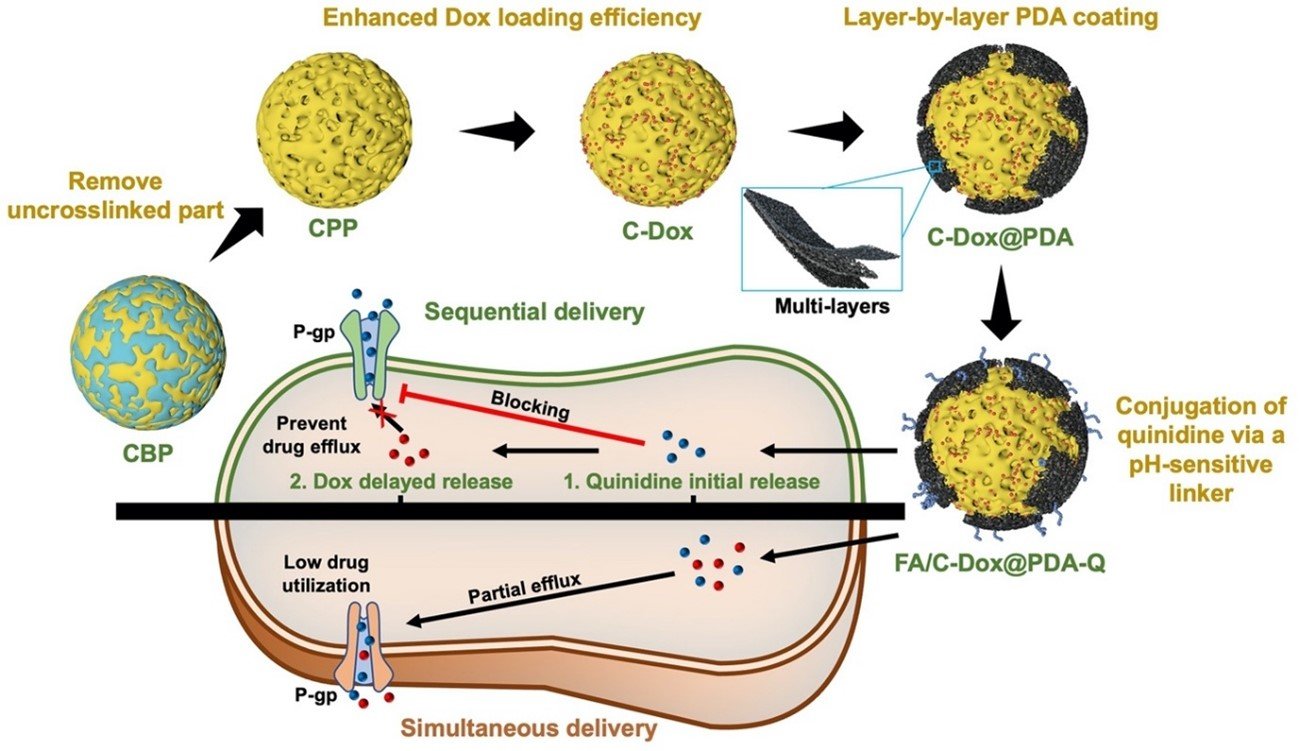
図1. 本研究の概念
<関連情報>
- https://www.tohoku.ac.jp/japanese/2026/05/press20260508-01-amino.html
- https://www.sciencedirect.com/science/article/pii/S0168365926003573
多剤耐性癌を克服するための逐次薬物送達用多機能アミノ酸ベースナノ粒子 Multifunctional amino acid-based nanoparticles for sequential drug delivery to overcome multidrug resistant cancer
Tengfei Wang, Nina Sang, Cécilia Ménard-Moyon, Eijiro Miyako, Alberto Bianco
Journal of Controlled Release Available online: 24 April 2026
DOI:https://doi.org/10.1016/j.jconrel.2026.114954
Highlights
- Porous amino acid nanoparticles are fabricated via a self-templating etching method.
- Polydopamine coating enables sequential release of quinidine prior to doxorubicin.
- Quinidine inhibits P-glycoprotein to reverse multidrug resistance in tumor cells.
- Folic acid conjugation provides active tumor targeting via folate receptor binding.
- Combined chemo/photothermal therapy achieves complete survival in drug-resistant mice.
Abstract
Multidrug resistance (MDR), often emerging after chemotherapy, has long restricted the therapeutic efficacy of cancer treatment in clinical practice. Among the various strategies developed to overcome MDR, the sequential delivery of P-glycoprotein (P-gp) inhibitors and anticancer drugs has shown great promise when compared with traditional co-delivery approaches. In this work, porous amino acid nanoparticles (NPs) are fabricated via a self-templating method and loaded with doxorubicin (Dox). NPs are coated with polydopamine (PDA), which is functionalized with quinidine, a P-gp inhibitor, via a pH-sensitive linker. The pH/glutathione (GSH) sensitivity of PDA allows a delayed release of Dox compared to quinidine, thereby enabling a sequential drug delivery. Specifically, the release profile of Dox enables to be precisely controlled through adjusting the number of PDA layers. In addition, the incorporation of PDA imparts photothermal capabilities to NPs, allowing for their use in synergistic photothermal therapy (PTT) and chemotherapy. Finally, the antitumor effect of NPs is evaluated in vitro and in vivo. The viability of MDR EMT-6/AR1 cells is decreased to less than 5% after the treatment with NPs and a significant tumor regression is observed in MDR tumor-bearing mice after combining PTT and chemotherapy, providing a high antitumor efficacy.