2024-04-08 インペリアル・カレッジ・ロンドン(ICL)
<関連情報>
- https://www.imperial.ac.uk/news/252415/long-covid-leaves-telltale-traces-blood/
- https://www.nature.com/articles/s41590-024-01778-0
入院後の長期COVID患者の大規模な表現型解析により、疾患のメカニズム的なサブタイプが明らかになる Large-scale phenotyping of patients with long COVID post-hospitalization reveals mechanistic subtypes of disease
Felicity Liew,Claudia Efstathiou,Sara Fontanella,Matthew Richardson,Ruth Saunders,Dawid Swieboda,Jasmin K. Sidhu,Stephanie Ascough,Shona C. Moore,Noura Mohamed,Jose Nunag,Clara King,Olivia C. Leavy,Omer Elneima,Hamish J. C. McAuley,Aarti Shikotra,Amisha Singapuri,Marco Sereno,Victoria C. Harris,Linzy Houchen-Wolloff,Neil J. Greening,Nazir I. Lone,Matthew Thorpe,A. A. Roger Thompson,PHOSP-COVID collaborative group & ISARIC investigators
Nature Immunology Published:08 April 2024
DOI:https://doi.org/10.1038/s41590-024-01778-0
Abstract
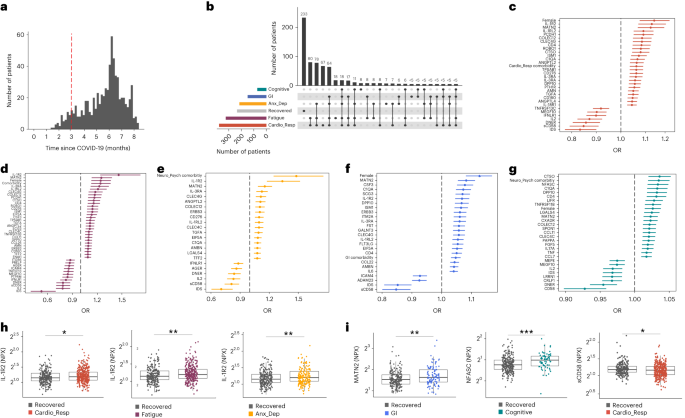
One in ten severe acute respiratory syndrome coronavirus 2 infections result in prolonged symptoms termed long coronavirus disease (COVID), yet disease phenotypes and mechanisms are poorly understood1. Here we profiled 368 plasma proteins in 657 participants ≥3 months following hospitalization. Of these, 426 had at least one long COVID symptom and 233 had fully recovered. Elevated markers of myeloid inflammation and complement activation were associated with long COVID. IL-1R2, MATN2 and COLEC12 were associated with cardiorespiratory symptoms, fatigue and anxiety/depression; MATN2, CSF3 and C1QA were elevated in gastrointestinal symptoms and C1QA was elevated in cognitive impairment. Additional markers of alterations in nerve tissue repair (SPON-1 and NFASC) were elevated in those with cognitive impairment and SCG3, suggestive of brain–gut axis disturbance, was elevated in gastrointestinal symptoms. Severe acute respiratory syndrome coronavirus 2-specific immunoglobulin G (IgG) was persistently elevated in some individuals with long COVID, but virus was not detected in sputum. Analysis of inflammatory markers in nasal fluids showed no association with symptoms. Our study aimed to understand inflammatory processes that underlie long COVID and was not designed for biomarker discovery. Our findings suggest that specific inflammatory pathways related to tissue damage are implicated in subtypes of long COVID, which might be targeted in future therapeutic trials.