2025-03-27 バーミンガム大学
<関連情報>
- https://www.birmingham.ac.uk/news/2025/smartwatches-may-help-control-diabetes-through-exercise
- https://bmjopen.bmj.com/content/15/3/e092260
2型糖尿病における運動と身体活動のアドヒアランスを強化するモバイルヘルスバイオメトリクス(MOTIVATE-T2D):英国とカナダで実施された分散型実現可能ランダム化比較試験 Mobile Health Biometrics to Enhance Exercise and Physical Activity Adherence in Type 2 Diabetes (MOTIVATE-T2D): a decentralised feasibility randomised controlled trial delivered across the UK and Canada
Katie Hesketh,Robert Andrews,Sandra Blitz,,Charlotte A Jones,Jonathan Little,Sarah L Percival,Richard Pulsford,Catherine L Russon,Joel Singer,Alison M McManus, Matthew Cocks
BMJ Open Published:March 26, 2025
DOI:10.1136/bmjopen-2024-092260

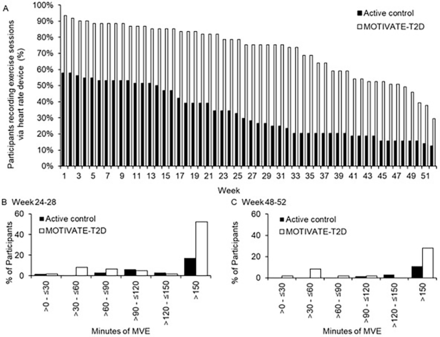
Figure 2
Exercise behaviour derived from optical heart rate monitoring. (A) Training drop-off by week in MOTIVATE-T2D and active control participants. (B) Proportion of participants achieving MVE duration at least once during weeks 24–28. (C) Proportion of participants achieving MVE duration at least once during weeks 48–52. MVE, moderate-to-vigorous intensity exercise; when calculating MVE, vigorous intensity exercise was multiplied by two. MOTIVATE-T2D, Mobile Health Biometrics to Enhance Exercise and Physical Activity Adherence in Type 2 Diabetes.
Abstract
Objectives Assess the feasibility of a mobile health (mHealth)-supported home-delivered physical activity (PA) intervention (MOTIVATE-T2D) in people with recently diagnosed type 2 diabetes (T2D).
Design Feasibility multicentre, parallel group, randomised controlled trial (RCT).
Setting Participants were recruited from England and Canada using a decentralised design.
Participants Adults (40–75 years) recently diagnosed with T2D (5–24 months).
Interventions Participants were randomised 1:1 to intervention (MOTIVATE-T2D) or active control groups. Participants codesigned 6month- home-delivered, personalised, progressive PA programmes supported by virtual behavioural counselling. MOTIVATE-T2D used biofeedback from wearable technologies to support the programme. The active control group received the same intervention without wearables.
Outcomes The primary outcomes were recruitment rate, retention and adherence to purposeful exercise. Clinical data on effectiveness were collected as exploratory outcomes at baseline, 6 and 12 months, with HbA1c and systolic blood pressure (BP) proposed as primary outcomes for a future full RCT.
Results n=135 eligible participants expressed an interest in the trial, resulting in 125 participants randomised (age 55±9 years, 48% female, 81% white), a recruitment rate of 93%. Retention at 12 months was 82%. MOTIVATE-T2D participants were more likely to start (OR 10.4, CI 3.4 to 32.1) and maintain purposeful exercise at 6 (OR 7.1, CI 3.2 to 15.7) and 12 months (OR 2.9, CI 1.2 to 7.4). Exploratory clinical outcomes showed a potential effect in favour of MOTIVATE-T2D, including proposed primary outcomes HbA1c and systolic BP (between-group mean differences: HbA1c: 6 months: -5% change from baseline, CI -10 to 2: 12 months: -2% change from baseline, CI -8 to -4; systolic BP: 6 months: -1 mm Hg, CI -5 to 3: 12 months: -4 mm Hg, CI -8 to 1).
Conclusions Our findings support the feasibility of delivering the MOTIVATE-T2D mHealth-supported PA intervention for people with recently diagnosed T2D and progression to a full RCT to examine its clinical and cost-effectiveness.
Trial registration number ISRCTN: 14335124; ClinicalTrials.gov: NCT0465353.