2026-03-08 中国科学院(CAS)
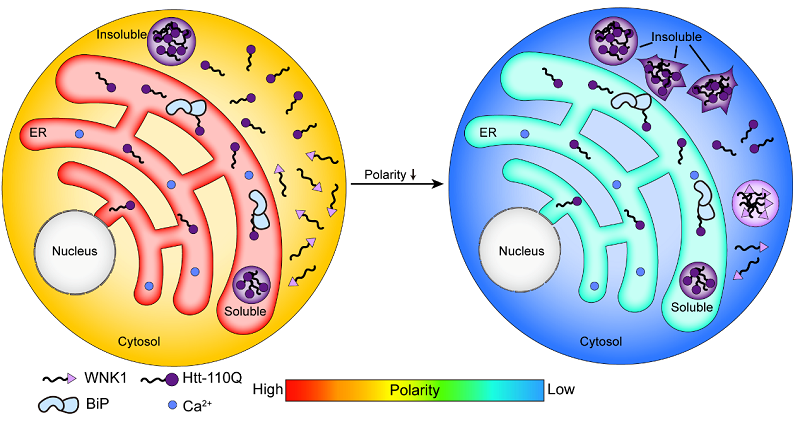
Schematic diagram of the model for polarity-regulated protein condensate and aggregate formation (Image by WANG Lei’s group)
<関連情報>
- https://english.cas.cn/newsroom/research-news/202603/t20260310_1152350.shtml
- https://www.nature.com/articles/s42003-025-09491-w
ヒト細胞において、小胞体は高い極性を示し、タンパク質凝集は少ない The endoplasmic reticulum displays high polarity with low protein aggregation in human cells
Xinwei Hu,Junlin Chen,Ping Liu,Huaiyue Zhang,Yu Liu,Xin Zhang & Lei Wang
Communications Biology Published:03 February 2026
DOI:https://doi.org/10.1038/s42003-025-09491-w
Abstract
Many physicochemical properties in the cellular milieu are important for cell function and survival. However, the polarity of different subcellular compartments and its role in protein condensate and aggregate formation within cells are less characterized. Here, we develop a method to compare the polarity in different subcellular compartments using the same polarity-sensitive solvatochromic fluorescent probe. Unexpectedly, the endoplasmic reticulum (ER) lumen displays a higher polarity and a more crowded environment than the cytosol in human cells. Polarity-decreasing and crowding-increasing hypertonic conditions induce condensate or aggregate formation of two intrinsically disordered proteins, with-no-lysine kinase 1 and Huntingtin gene (Htt) exon1 with an expanded polyQ stretch (Htt-polyQ), in the cytosol. However, targeting Htt-polyQ to the ER prevents its aggregation, suggesting that polarity but not crowding is more relevant to protein aggregation. Our results reveal the heterogeneity in subcellular polarity and crowding, and uncover previously unrecognized high-polarity in the ER lumen, which provides a unique environment for maintaining robust proteostasis.