2026-03-13 東北大学
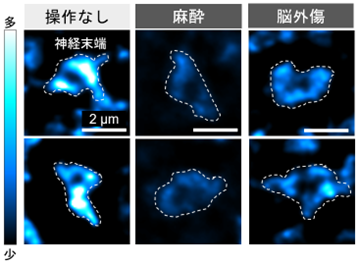
図1. 麻酔や脳外傷によってシナプスで起こる変化。貯蔵プールを構成する分子シナプシンを蛍光標識した。操作により、シナプシンの量が減少した。
<関連情報>
- https://www.tohoku.ac.jp/japanese/2026/03/press20260313-01-memory.html
- https://www.tohoku.ac.jp/japanese/newimg/pressimg/tohokuuniv-press20260313_01web_memory.pdf
- https://www.pnas.org/doi/10.1073/pnas.2514875123
逆行性健忘における選択的小胞プールの破壊はシナプス前終末における記憶を解離させる Disruption of a selective vesicle pool upon retrograde amnesia dissociates memory at presynaptic terminals
Shun Hiramatsu, Kaito Kabetani, Shu Kondo, and Hiromu Tanimoto
Proceedings of the National Academy of Sciences Published:March 5, 2026
DOI:https://doi.org/10.1073/pnas.2514875123
Significance
What determines memory stability? We show that distinct presynaptic vesicle populations underlie the labile and consolidated memories, which are simultaneously formed following odor-shock associative learning in fruit flies. These distinct synaptic mechanisms explain the selective loss of labile memory triggered by traumatic perturbations after learning. Given the evolutionarily conserved regulators of vesicle dynamics, our findings provide insights into memory consolidation across species.
Abstract
A single learning episode induces both labile and consolidated forms of aversive olfactory memory in Drosophila melanogaster. Retrograde amnesia triggered by post-learning perturbations specifically impairs the labile memory. However, synaptic mechanisms for this selectivity remain elusive. Here, we show that diverse amnestic treatments, such as concussion, commonly disrupt the presynaptic clustering of Synapsin, which is required for anesthesia-sensitive memory. Consistently, targeted knockout of synaptojanin, a key regulator of the endocytic pathway, selectively impaired labile memory and the Synapsin-associated vesicles. In contrast, we identified Rab3, a small GTPase that regulates the late steps of vesicle exocytosis at the active zone, as selectively required for anesthesia-resistant memory. Rab3 hyperactivation enhanced its association with vesicles while displacing Synapsin. Strikingly, this manipulation biased memory toward stabilization at the cost of the labile component. We thus propose distinct vesicle pools at the presynaptic terminal underlie the formation of labile and consolidated memories. Thus, our work offers a molecular framework for controlling memory stability through targeted manipulation of vesicle dynamics.