2026-03-24 京都大学
<関連情報>
- https://www.kyoto-u.ac.jp/ja/research-news/2026-03-24
- https://www.kyoto-u.ac.jp/sites/default/files/2026-03/web_2603_Takauchi-76d7373d2d5a25ef9c14e6e4719d5a30.pdf
- https://www.nature.com/articles/s41590-026-02447-0
組織特異的な二次的TCR刺激が、幹細胞様Th17細胞からCD200陽性Egr2高発現関節炎惹起性Th17細胞への表現型転換を誘導する Tissue-restricted secondary TCR engagement drives the transition from stem-like to CD200+ Egr2hi arthritogenic Th17 cells
Yusuke Takeuchi,Daiya Ohara,Hitomi Watanabe,Yuji Nishimura,Takeshi Iwasaki,Shohei Hori,Hiroshi Kawamoto,Hiroki Kato,Gen Kondoh,Akio Morinobu,Tsuneyo Mimori & Keiji Hirota
Nature Immunology Published:23 March 2026
DOI:https://doi.org/10.1038/s41590-026-02447-0
Abstract
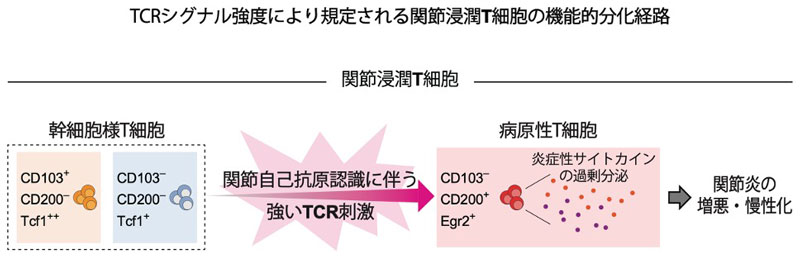
Excessive activation of interleukin-17-producing helper T (TH17) cells can cause autoimmune tissue inflammation. However, how TH17 cells enhance their pathogenicity within target tissues and whether destabilized regulatory T cells contribute to pathogenic TH17 cell populations remain unclear. Using a TH17 cell-dependent autoimmune arthritis model, we demonstrated that TH17 and regulatory T cells did not undergo significant mutual plasticity, based on lineage-tracing and T cell receptor (TCR) repertoire analyses. Single-cell RNA sequencing of joint CD4+ T cells revealed three phenotypically distinct TH17 clusters, ranging from a CD103⁺ Tcf1hi stem-like state to a CD200⁺ Egr2hi highly pathogenic state. The phenotypic transition to the CD200⁺ pathogenic state was not a default progression driven by inflammatory cues, but rather a highly selective process mediated by tissue-restricted secondary TCR engagement within inflamed joints. Our findings delineate the heterogeneity and pathogenic potential of arthritogenic TH17 cells, highlighting secondary autoimmune TCR signaling as a critical regulatory determinant of their developmental trajectories that may serve as a therapeutic target for autoimmune arthritis.