2026-03-23 国立がん研究センター,神戸大学
<関連情報>
- https://www.ncc.go.jp/jp/information/pr_release/2026/0323/index.html
- https://ascopubs.org/doi/10.1200/JCO.2025.43.16_suppl.6007
アンドロゲン受容体陽性唾液腺癌に対するダロルタミドとゴセレリンの併用療法:第2相試験(DISCOVARY)の結果。 Darolutamide plus goserelin for androgen receptor-positive salivary gland cancers: Results of phase 2 study (DISCOVARY).
Susumu Okano, Makoto Tahara, Kiyoaki Tsukahara, Tomoyuki Otsuka, Satoshi Kano, Masato Nagaoka, Hideoki Uryu, … , and Naomi Kiyota
Journal of Clinical Oncology Published:May 28, 2025
DOI:https://doi.org/10.1200/JCO.2025.43.16_suppl.6007
Abstract
Background: No standard treatment exists for unresectable locally advanced (LA) or recurrent/metastatic (R/M) salivary gland cancer (SGC). Previous findings suggest that combined androgen blockade (CAB) provides promising clinical activity in patients with androgen receptor (AR)-positive SGC. However, no AR-targeted drug is currently approved for SGC. This multi-center phase 2 study investigated two approaches in patients with unresectable LA or R/M SGC: darolutamide monotherapy followed by the combination of darolutamide and goserelin. In the monotherapy phase, darolutamide showed an objective response rate (ORR) of 20.8% as determined by independent central review (ICR) with tolerable toxicity (ASCO 2023). We now report the results from the combination phase.
Methods: Eligible patients had histologically confirmed AR-positive LA or R/M SGC, ECOG performance status (PS) 0–2, adequate organ function, and no local therapy options. Patients received darolutamide orally at 1,200mg daily, combined with goserelin at 3.6 mg every four weeks. The primary endpoint was ORR by ICR in patients verified to have AR positivity through central assessment. Secondary endpoints included clinical benefit rate (CBR), disease control rate (DCR), progression-free survival (PFS), overall survival (OS), and safety profiles.
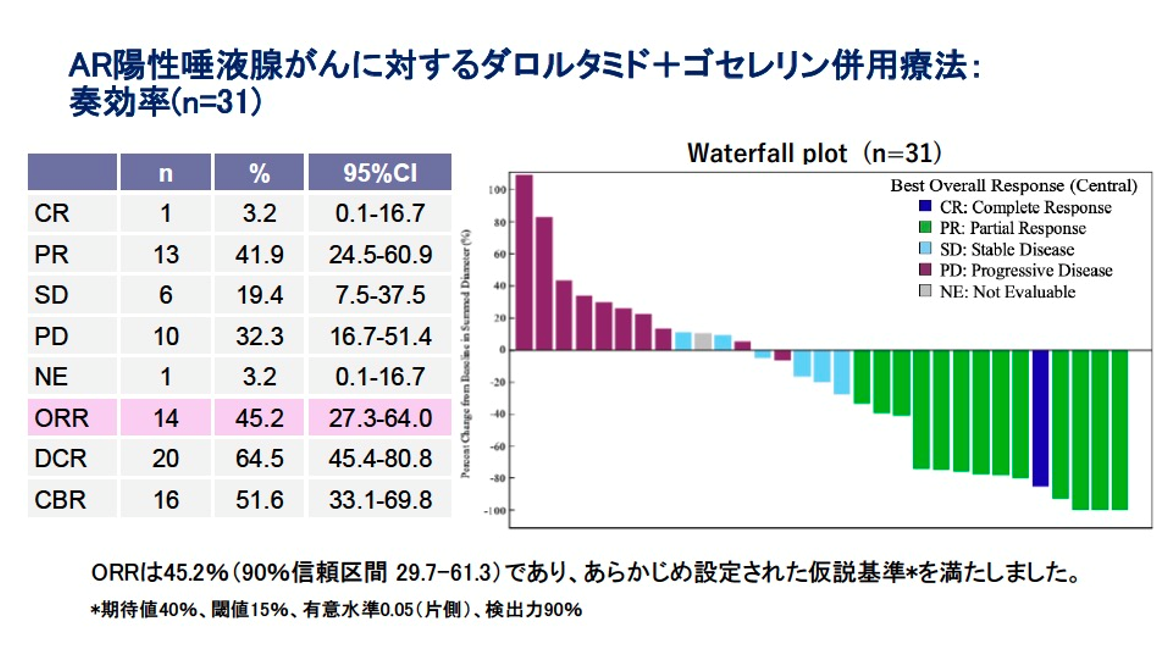
Results: Between Sep 2022 and Aug 2023, 33 patients were enrolled in the combination phase. Median age was 63 years; 26 were male; ECOG PS 0/1/2 in 28/4/1. Histology included salivary duct carcinoma (n=32) or adenocarcinoma not otherwise specified (n=1). Prior treatment included surgery (n=23), radiotherapy (n=21), and chemotherapy (n=15). The ORR by ICR was 45.2% (14/31; 95% CI, 27.3–64.0), meeting the primary endpoint. CBR was 51.6% (95% CI, 33.1–69.8), and DCR was 64.5% (95% CI, 45.4–80.8). At a median follow-up of 13.7 months, the median PFS was 13.1 months (95% CI, 2.0– not calculable [NC]). Thirteen patients continued treatment at the data cutoff (August 9, 2024). Median OS was not reached (95% CI, 20.0–NC), and 12 months OS rate was 87%(95% CI, 68.9–94.9). Treatment was generally well tolerated, with six patients (18.2%) experiencing grade 3 adverse events.
Conclusions: This is the first prospective CAB trial in SGC which has met its primary endpoint. Darolutamide plus goserelin demonstrated clinically meaningful efficacy and a favorable safety profile, suggesting it may be a compelling option before initiating chemotherapy, which can significantly diminish a patient’s quality of life. Clinical trial information: NCT05694819.