2026-03-27 中国科学院(CAS)
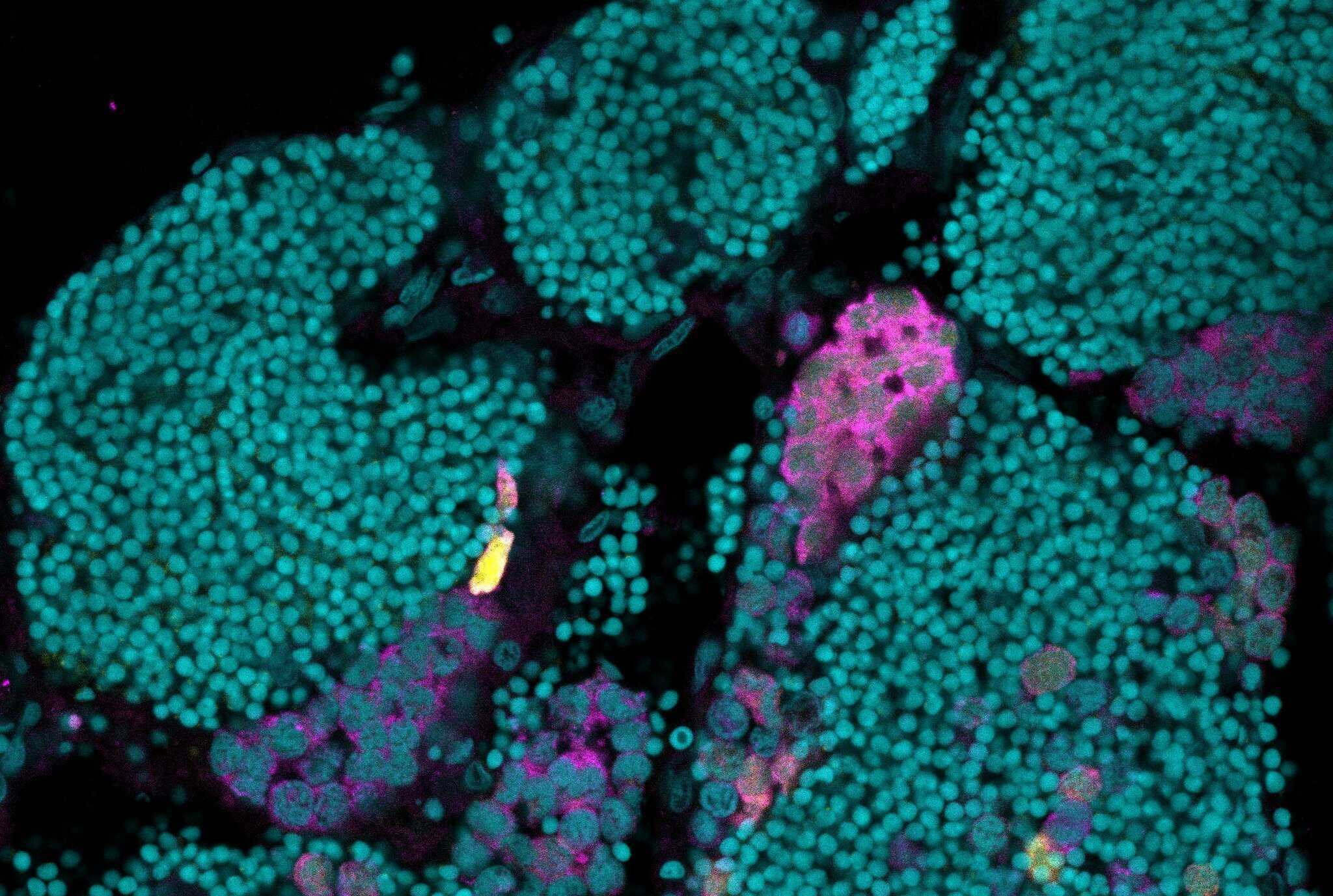
Engineering iTreg-mediated immune tolerance in zebrafish overcomes allograft rejection. (Image by IHB)
<関連情報>
- https://english.cas.cn/newsroom/research-news/202603/t20260327_1154083.shtml
- https://www.sciencedirect.com/science/article/pii/S1673852726000688
ゼブラフィッシュにおける同種移植の障壁を回避するための、foxp3a過剰発現によるTregを介した免疫寛容の誘導 Engineering Tregs-mediated immune tolerance via foxp3a overexpression to evade allograft transplantation barriers in zebrafish
Junwen Zhu, Yongkang Hao, Fenghua Zhang, Xiaxia Gao, Houpeng Wang, Liqun Yu, Xiaosi Wang, Yonghua Sun
Journal of Genetics and Genomics Available online: 2 March 2026
DOI:https://doi.org/10.1016/j.jgg.2026.02.024
Abstract
In mammals, regulatory T cells (Tregs) are widely exploited to promote immune tolerance in organ transplantation. In zebrafish, although germline stem cell (GSC) or gonadal primordium transplantation into immunodeficient hosts can accelerate gamete production, maintaining immunocompromised lines presents substantial practical challenges. To overcome this limitation, this study generates a Tg(CMV:foxp3a) zebrafish line through systemic overexpression of Forkhead box P3a (Foxp3a), the lineage-defining transcription factor of Tregs. Transcriptomic and in situ hybridization analysis reveal downregulation of the Treg negative regulator cd127 and upregulation of multiple immunosuppressive factors in the head kidney and thymus. Single-cell RNA sequencing further demonstrates a reduction in effector T and B cell populations, accompanied by an increase in quiescent T cells exhibiting resting Treg-like features. Importantly, using Tg(CMV:foxp3a) fish as hosts for subcutaneous gonadal primordium transplantation (SGPT) and intraperitoneal GSC transplantation (IGCT) markedly accelerates germ cell maturation and enables efficient establishment of stable transgenic lines. Post-transplantation analysis indicates delayed and attenuated immune activation, enhanced graft survival, and rapid induction of immunosuppressive states. Together, foxp3a overexpression reshapes the immune landscape to confer immune tolerance, providing a practical Tregs-based alternative to immunodeficient hosts for fish genome manipulation and transplantation.