2026-04-07 スイス連邦工科大学ローザンヌ校(EPFL)
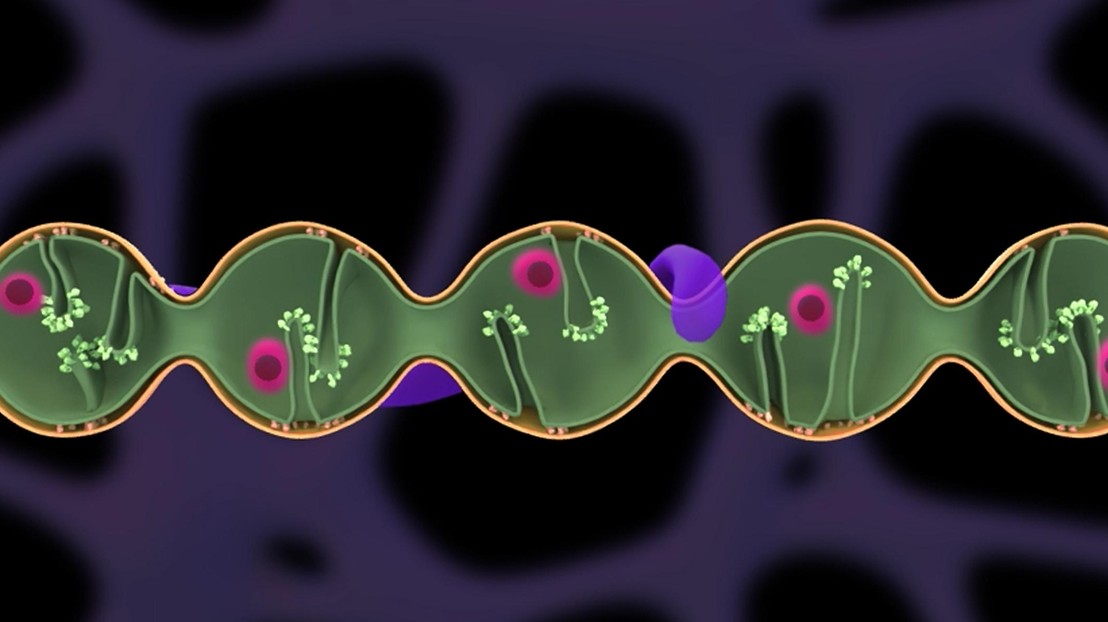
Pearling in a mitochondrion. Credit: Suliana Manley © 2026 EPFL
<関連情報>
- https://actu.epfl.ch/news/how-mitochondria-organize-our-second-genome/
- https://www.science.org/doi/10.1126/science.adu5646
パール形成がミトコンドリアDNAヌクレオイドの分布を促進する Pearling drives mitochondrial DNA nucleoid distribution
Juan C. Landoni, Matthew D. Lycas, Josefa Macuada, Willi Stepp, […] , and Suliana Manley
Science Published:2 Apr 2026
DOI:https://doi.org/10.1126/science.adu5646
Editor’s summary
Mitochondria contain their own DNA packaged into structures called nucleoids, which must be evenly spaced along the organelle’s network to ensure function and inheritance. Landoni et al. found that this spacing arises from “pearling,” a physical instability that transforms a mitochondrion from a tubule into regularly spaced, connected beads (see the Perspective by van den Ameele and Prudent). Pearling occurs reversibly in response to calcium influx, carrying nucleoids along, splitting them, and leaving them spaced at regular intervals. The frequency and duration of pearling are defined by the dynamic inner mitochondrial membrane architecture, linking mitochondrial physiology to genome organization. —Stella M. Hurtley
Abstract
The distribution of mitochondrial DNA–containing nucleoids is essential for mitochondrial function and genome inheritance; however, no known mechanisms can explain nucleoid segregation or their regular positioning. In this work, we found that mitochondria frequently undergo a reversible biophysical instability termed “pearling,” transforming from a tubular into a regularly spaced beads morphology. Physiological pearling imposed a characteristic length scale and simultaneously mediated nucleoid disaggregation and established internucleoid distancing with high precision. Pearling onset was triggered by calcium influx, whereas the density of lamellar cristae invaginations modulated pearling prevalence and preserved nucleoid spacing following recovery. The dysregulation of mitochondrial calcium influx or inner membrane cristae integrity caused aberrant nucleoid clustering. Our results identify pearling as a mechanism governing nucleoid distribution and inheritance and offer insights into its regulation.