2026-04-27 バーミンガム大学
<関連情報>
- https://www.birmingham.ac.uk/news/2026/obesity-leaves-a-lasting-memory-in-immune-cells
- https://link.springer.com/article/10.1038/s44319-026-00765-w
CD4 Tリンパ球における肥満のDNAメチル化を介した記憶は免疫調節異常を永続させる DNA methylation-mediated memory of obesity in CD4 T lymphocytes perpetuates immune dysregulation
Jennifer Niven,Salih Kucuk,Atrayee Gope,Michelangelo Certo,Fearon C Cassidy,Ainhoa Arana Echarri,Sadaf Ali,Efthymios Ladoukakis,Sofia Vidali,Chiara Macchi,Sayeda S Amir,Ronan Bergin,Sophie Davies,Oliver J Perkin,Joanne Smith,Danilo Cucchi,Helen Heneghan,Susanne Wijesinghe,Benjamin J Jenkins,Shanat Baig,Christopher Mahony,Chiamaka Chidomere,Sovan Sarkar,Anna Nicolaou,… Claudio Mauro
EMBO Reports Published:27 April 2026
DOI:https://doi.org/10.1038/s44319-026-00765-w
Abstract
Obesity represents a major global healthcare crisis, with childhood obesity rising at an alarming rate. Children with obesity are highly likely to carry it into adulthood, bringing numerous associated health risks. Even more troubling is the emerging understanding of “obesity memory”, which contributes to the frequent issue of weight regain. Here, we show that obesity imprints CD4 T cells through DNA methylation, leading to a long-time lag, spanning years, before adaptive immune homeostasis is restored after weight loss. Differential DNA methylation analysis highlights autophagy and immune senescence as potential key mechanisms underpinning this memory of obesity in CD4 T cells. In addition, particularly palmitate could be a key saturated fatty acid that can contribute to epigenetic alterations in CD4 T cells, potentially perpetuating this altered state. We identify molecular candidates (i.e., Stk26 and Cdkn1c) underpinning key cell functions (autophagy and immune senescence) that could be targeted to promote a return to immune homeostasis alongside weight loss. These findings raise the possibility that targeting such pathways could support the restoration of immune homeostasis alongside weight loss therapies.
Synopsis
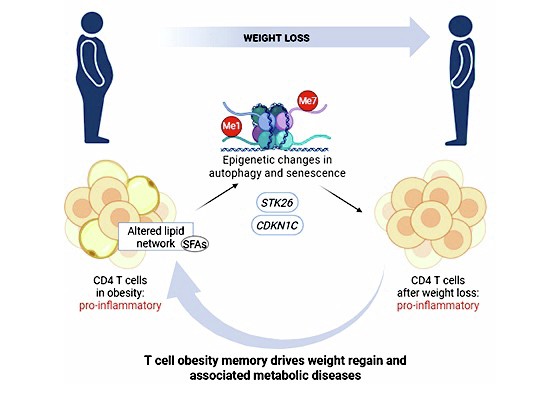
Adaptive immunity has a long-lasting memory of obesity via DNA methylation as key mechanism. Autophagy and immune senescence are two pathways implicated in this memory that could be targeted to help restore immune homeostasis alongside weight loss therapies.
- Obesity leaves a lasting memory in CD4 T cells, persisting after weight loss.
- This memory is driven by DNA methylation in CD4 T cells.
- Autophagy and immune senescence drive obesity memory in CD4 T cells.
- Sustained long-term weight loss is crucial to restore adaptive immune homeostasis.