2026-04-24 中国科学院(CAS)
<関連情報>
- https://english.cas.cn/newsroom/cas-in-media/202604/t20260424_1157941.shtml
- https://www.science.org/doi/10.1126/science.adw2156
生体内機能獲得型Perturb-seqを用いたアストロサイトにおける転写因子機能のマッピング Mapping transcription factor functions in astrocytes using in vivo gain-of-function Perturb-seq
Liansheng Zhang, Qi Ma, Xiangrui Kong, Weijuan Zou, […] , and Haibo Zhou
Science Published:23 Apr 2026
DOI:https://doi.org/10.1126/science.adw2156
Editor’s summary
Astrocytes play an important role in regulating brain functions and are involved in several neurological disorders. Zhang et al. investigated genetic factors affecting astrocyte biology by developing an in vivo gain-of-function Perturb-seq approach. This method was used to map astrocytic transcription factors in physiological and pathological conditions in mice. In a model of neuroinflammation, the authors identified a transcription factor that may be a potential therapeutic candidate for Alzheimer’s disease. These results unveil aspects of astrocyte biology in health and disease. —Mattia Maroso
Structured Abstract
INTRODUCTION
Perturb-seq enables high-throughput profiling of the effects of genetic perturbation across many genes simultaneously, revealing their regulatory networks and functional roles. Extending this approach in vivo with cell type–specific readouts allows systematic dissection of genotype-phenotype relationships within tissues. Astrocytes, which are essential for brain homeostasis, synapse maintenance, and neuroinflammation, are tightly regulated by transcription factors (TFs). However, the landscape of TF-mediated regulation in astrocytes in vivo, across both physiological and pathological states, remains poorly understood.
RATIONALE
Recent in vivo single-cell perturbation methods enable gene function studies in living tissues but remain limited to small-scale loss-of-function analyses under physiological conditions, limiting therapeutic exploration. We developed an in vivo gain-of-function platform (iGOF–Perturb-seq) that integrates barcoded open reading frames, adeno-associated virus (AAV)–mediated delivery, and single-nucleus RNA sequencing (snRNA-seq) to systematically assess gene overexpression effects of perturbing a large number of genes in living tissues. Applying this platform to astrocytes in the mouse brain, we profiled GOF effects for ~1000 TFs, uncovering regulatory networks that modulate astrocyte functions in complex biological contexts. We further extended the platform to mice that model disease conditions, enabling screening of therapeutic targets in vivo.
RESULTS
We constructed an AAV-mediated overexpression library of ~1000 TFs and applied the iGOF–Perturb-seq platform with targeted GOF and loss-of-function validations to create a functional atlas of astrocyte TFs. This large-scale analysis provides a valuable resource for understanding how TFs regulate gene networks and cellular functions in vivo. Applying gene expression program analysis for the transcriptomic profiles of perturbed astrocytes, we identified cofunctional TF modules and predicted functions of previously uncharacterized TFs, uncovering transcriptional networks that define astrocyte functions in vivo. To further elucidate TF-mediated transcriptional networks, we performed high-dimensional weighted gene coexpression network analysis across all perturbed astrocytes. This analysis resolved five coexpression modules enriched for distinct biological processes and yielded six clusters of TFs, revealing how diverse GOF TF perturbations could produce converging effects on functionally transcriptional programs in astrocytes. By comparing our Perturb-seq data with snRNA-seq datasets from normal aged subjects and patients with neurodegenerative diseases, glioma, and psychiatric disorders, we predicted potential disease-associated TFs that may contribute to disease progression and successfully validated their functional roles. More importantly, Perturb-seq under simulated disease conditions enabled direct analysis of gene regulation in pathological conditions. For example, we identified astrocyte-specific Ferd3l as a potential therapeutic candidate for neurodegenerative diseases. Brain-wide overexpression of Ferd3l not only suppressed inflammatory responses and neurotoxic activation of astrocytes but also alleviated cognitive deficits and reactive astrocyte-mediated neurotoxicity in an Alzheimer’s disease (AD) mouse model, demonstrating both its neuroprotective potential and the utility of iGOF–Perturb-seq for systematic in vivo screening of therapeutic targets.
CONCLUSION
Our study establishes a functional atlas of TFs in astrocytes using iGOF–Perturb-seq under both physiological and pathological conditions, providing a valuable resource for understanding gene regulation and molecular mechanisms underlying astrocyte functions in healthy and diseased brains, as well as for identifying potential pathogenic and therapeutic targets across diverse brain disorders.
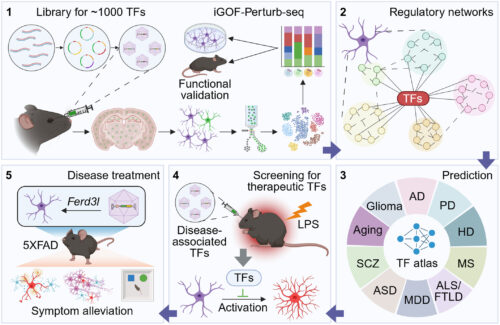
Building a functional atlas of TFs in astrocytes using in vivo GOF Perturb-seq under physiological and pathological conditions.
(1) Development of an iGOF–Perturb-seq platform to investigate TF functions in astrocytes. (2) Elucidation of TF regulatory roles across diverse biological processes in astrocytes. (3) Prediction of disease-associated TFs for diverse brain diseases based on the TF atlas. (4) Screening of therapeutic TFs under pathological conditions using iGOF–Perturb-seq. (5) Treatment of AD-related pathologies in 5XFAD mice through AAV-mediated, brain-wide, astrocyte-specific overexpression of Ferd3l. ORF, open reading frame; FACS, fluorescence-activated cell sorting; LPS, lipopolysaccharide; AD, Alzheimer’s disease; PD, Parkinson’s disease; HD, Huntington’s disease; MS, multiple sclerosis; ALS/FTLD, amyotrophic lateral sclerosis/frontotemporal lobar degeneration; MDD, major depressive disorder; ASD, autism spectrum disorder; SCZ, schizophrenia.
Abstract
An in vivo approach combining high-throughput screening with cell type–specific readouts could enable elucidation of genotype-phenotype relationships in complex tissues. We developed an in vivo gain-of-function Perturb-seq platform, termed iGOF–Perturb-seq, to build a functional atlas of ~1000 transcription factors (TFs) in astrocytes, a cell type essential to many brain functions. We then identified cofunctional modules, annotated uncharacterized TFs, and predicted disease-associated TF clusters. Furthermore, iGOF–Perturb-seq performed in a mouse neuroinflammatory model identified Ferd3l as a therapeutic candidate, and astrocyte-specific overexpression of Ferd3l alleviated Alzheimer’s disease symptoms in mice. This study provides resources for understanding gene regulation and disease mechanisms in vivo and for identifying potential therapeutic targets for different brain diseases.