2022-05-17 アメリカ国立標準技術研究所(NIST)
研究者たちは、既存の方法をベースに、効率、アクセス性、品質管理を改善しました。新しい動物実験不要のプロトコルを用いて92の化学物質を評価したところ、77%の化合物について一般的な動物実験法と同じ結果が得られました。この結果は本日発行のToxics誌に掲載され、新しい測定方法の信頼性を高め、国際貿易の拡大と動物実験の削減を両立する標準化への道を開く可能性があります。
動物を用いない試験に対する信頼性を高め、その採用を促進するため、国立環境衛生科学研究所の常設委員会がコーディネートしたいくつかの連邦機関が、新しい有望な方法の信頼性を評価している。委員会のメンバー機関であるCPSCは、NISTと提携し、親電子アレルゲン・スクリーニング・アッセイ(EASA)として知られるDPRAよりはるかに高速な方法を評価しています。
<関連情報>
- https://www.nist.gov/news-events/news/2022/05/nist-study-gives-animal-testing-alternatives-confidence-boost
- https://www.mdpi.com/2305-6304/10/5/257
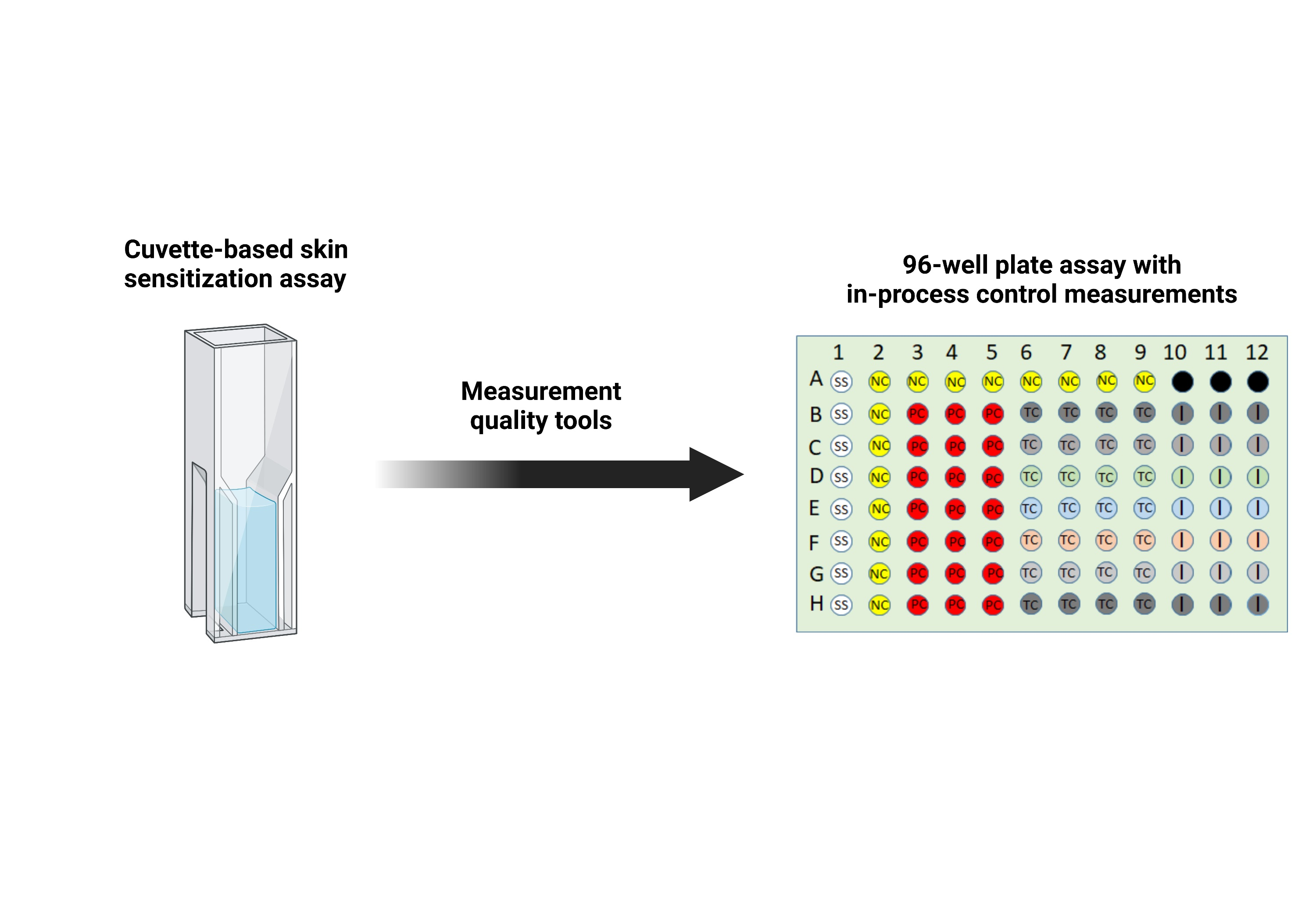
計測科学的アプローチによる皮膚感作のための96ウェル親水性アレルゲンスクリーニングアッセイの開発 Development of a 96-Well Electrophilic Allergen Screening Assay for Skin Sensitization Using a Measurement Science Approach
Elijah J. Petersen,Richard Uhl,Blaza Toman,John T. Elliott,Judy Strickland,James Truax and John Gordon
Toxics Published: 17 May 2022
DOI:https://doi.org/10.3390/toxics10050257
Abstract
The Electrophilic Allergen Screening Assay (EASA) has emerged as a promising in chemico method to detect the first key event in the adverse outcome pathway (AOP) for skin sensitization. This assay functions by assessing the depletion of one of two probe molecules (4-nitrobenzenethiol (NBT) and pyridoxylamine (PDA)) in the presence of a test compound (TC). The initial development of EASA utilized a cuvette format resulting in multiple measurement challenges such as low throughput and the inability to include adequate control measurements. In this study, we describe the redesign of EASA into a 96-well plate format that incorporates in-process control measurements to quantify key sources of variability each time the assay is run. The data from the analysis of 67 TCs using the 96-well format had 77% concordance with animal data from the local lymph node assay (LLNA), a result consistent with that for the direct peptide reactivity assay (DPRA), an OECD test guideline (442C) protein binding assay. Overall, the measurement science approach described here provides steps during assay development that can be taken to increase confidence of in chemico assays by attempting to fully characterize the sources of variability and potential biases and incorporate in-process control measurements into the assay.