2026-03-20 デューク大学
<関連情報>
- https://pratt.duke.edu/news/oct-ai-wound-monitoring/
- https://www.cell.com/cell-biomaterials/fulltext/S3050-5623(26)00078-4
深層学習を用いたマルチモーダルOCTにより、ハイドロゲル処理された創傷における生体内治癒動態が明らかになる Multimodal OCT with deep learning reveals in vivo healing dynamics in hydrogel-treated wounds
Jiyeon Song ∙ Shreyas Shah ∙ Makenzie Bushold ∙ … ∙ Ya Guan ∙ Michael S. Eggleston ∙ Sharon Gerecht
Cell Biomaterials Published:March 20, 2026
DOI:https://doi.org/10.1016/j.celbio.2026.100422
Graphical abstract

The bigger picture
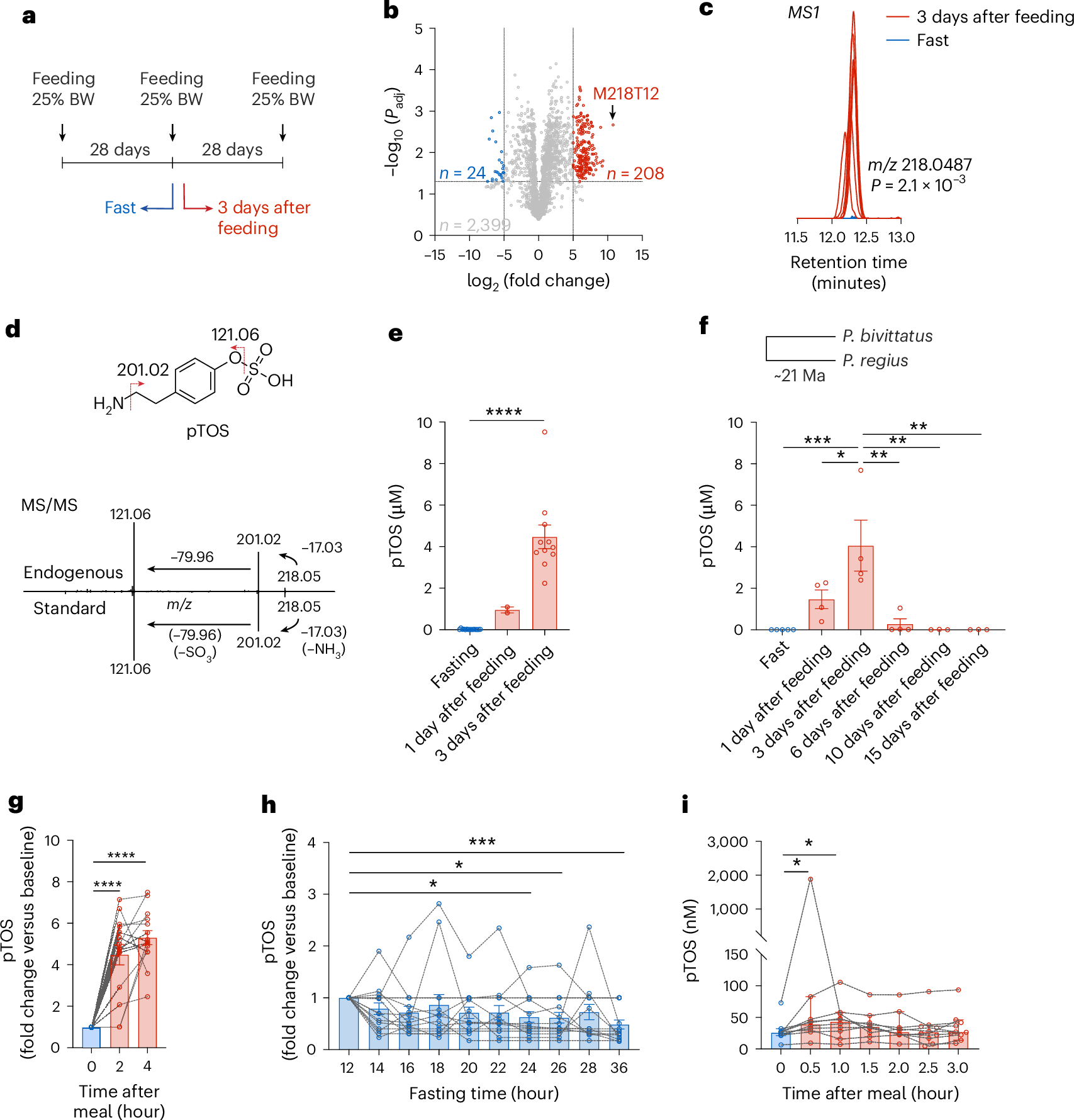
Chronic wounds, including diabetic ulcers, burns, and surgical wounds, affect millions of patients worldwide, imposing substantial burdens on healthcare systems and quality of life. Despite advances in wound care, clinicians still rely primarily on surface-level assessments such as photography and wound size measurements, which fail to capture the subsurface biological processes that ultimately determine healing outcomes. The inability to non-invasively monitor tissue regeneration longitudinally delays early detection of healing complications.
Biomaterial-based approaches, particularly hydrogels, have shown remarkable promise as therapeutic platforms for wound healing. Their tunable mechanical properties allow precise modulation of the wound microenvironment, influencing key biological processes such as immune cell polarization, vascular remodeling, and matrix deposition. However, understanding how these material properties dynamically regulate tissue regeneration in vivo has been hindered by the lack of tools that can capture these interactions longitudinally and non-destructively.
This work addresses both challenges simultaneously. By integrating optical coherence tomography (OCT) with AI-based analytics, we enable non-invasive, depth-resolved longitudinal tracking of tissue compartments and vascular remodeling. Applied to dextran-based hydrogels of tunable stiffness, this platform reveals how mechanical properties shape distinct healing trajectories, linking material design to biological outcomes in a quantitative and reproducible manner.
Looking ahead, this integrated multimodal OCT-AI platform has broad implications. It offers a powerful approach to rational biomaterial design, enabling researchers to screen and optimize material properties based on dynamic in vivo responses rather than endpoint measurements alone. Clinically, such a platform could transform wound monitoring by providing objective, data-driven assessments that guide therapeutic decisions and improve patient outcomes across a range of wound types.
Highlights
- Multimodal OCT-AI provides objective, quantitative metrics for wound assessment
- Automated AI-driven 3D segmentation tracks eight distinct tissue subclasses
- qSV-OCT captures depth-resolved vascular remodeling without contrast agents
- Stiffer hydrogels promote earlier immune resolution and accelerated healing
Summary
Monitoring hydrogel-guided wound healing with high spatial resolution without invasive procedures remains a key clinical challenge. We developed an integrated platform combining optical coherence tomography (OCT) with artificial intelligence (AI) to monitor biomaterial-modulated tissue regeneration quantitatively. Using AI-assisted 3D segmentation and enhanced speckle variance OCT, we tracked longitudinal changes in granulation tissue, dermal restoration, and vascular remodeling in wounds treated with hydrogels of tunable stiffness. This multimodal OCT-AI platform enabled quantitative characterization of stiffness-dependent differences in wound healing progress and trajectories. Stiff hydrogels promoted earlier vascular remodeling and accelerated healing, whereas soft hydrogels exhibited delayed phase transitions and slower overall healing. These OCT-derived measurements were well-aligned with histological and immunofluorescence analyses, confirming earlier inflammation resolution in stiff hydrogel-treated wounds and validating the accuracy and sensitivity of this non-invasive approach. This multimodal OCT-AI platform shows strong potential for preclinical treatment evaluation and clinical wound monitoring.