2026-03-25 アメリカ国立衛生研究所(NIH)
<関連情報>
- https://www.nih.gov/news-events/news-releases/chronic-inflammation-leaves-long-lasting-impression-gut-stem-cells-increasing-colorectal-cancer-risk
- https://www.nature.com/articles/s41586-026-10258-4
大腸炎のエピジェネティック記憶は腫瘍の増殖を促進する Epigenetic memory of colitis promotes tumour growth
Surya Nagaraja,Lety Ojeda-Miron,Ruochi Zhang,Ena Oreskovic,Conrad Hock,Yan Hu,Daniel Zeve,Karina Sharma,Roni R. Hyman,Qiming Zhang,Andrew Castillo,David T. Breault,Ömer H. Yilmaz & Jason D. Buenrostro
Nature Published:25 March 2026
DOI:https://doi.org/10.1038/s41586-026-10258-4
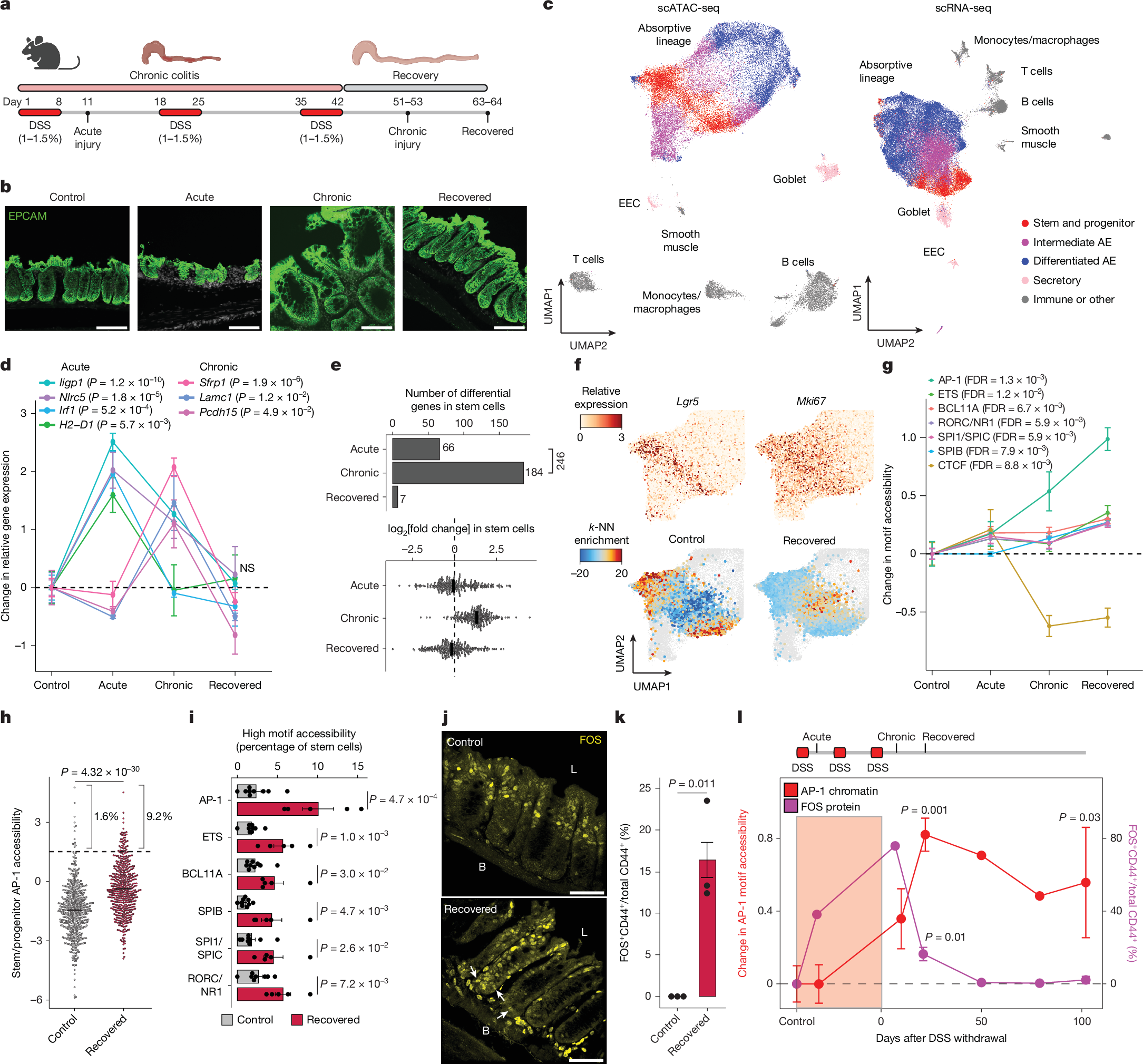
Abstract
Chronic inflammation is a well-established risk factor for cancer, but the underlying molecular mechanisms remain unclear1,2. Using a mouse model of colitis, we demonstrate that colonic stem cells retain an epigenetic memory of inflammation following disease resolution that persists for more than 100 days. Here we find that memory of colitis is characterized by a cumulative gain of activator protein 1 (AP-1) transcription factor activity, with durable changes to chromatin accessibility. Further, we develop SHARE-TRACE, a method that enables simultaneous profiling of gene expression, chromatin accessibility and clonal history in single cells, enabling high-resolution tracking of epigenomic memory. This approach reveals that memory of colitis is propagated cell-intrinsically and inherited through stem cell divisions, with some clones demonstrating stronger memory than others. Finally, we show that colitis primes stem cells for increased expression of an AP-1-regulated gene program following oncogenic mutation that accelerates tumour growth, a phenotype dependent on AP-1 activity. Together, our findings provide a mechanistic link between chronic inflammation and malignancy, revealing how long-lived epigenetic alterations in regenerative tissues may contribute to disease susceptibility and suggesting potential diagnostic and therapeutic strategies to mitigate cancer risk in patients with chronic inflammatory conditions.


