2023-05-31 カリフォルニア大学リバーサイド校(UCR)
◆これらの細菌は、化学構造内に1つ以上の塩素原子を持つ一群のパーフルオロアルキル物質(PFAS)を分解します。これらの有害な永遠の化学物質は、非常に強い炭素-フッ素結合のために環境中で何十年もの間残留します。UCRチームは、細菌が汚染物質の塩素-炭素結合を切断し、これによって永遠の化学物質の構造を破壊し、無害化する反応を開始することを発見しました。この研究は、生物処理が化学処理よりも費用がかからず、環境に優しいため、有望な方法であることを示しています。
<関連情報>
- https://news.ucr.edu/articles/2023/05/31/biological-cleanup-discovered-certain-forever-chemicals
- https://pubs.acs.org/doi/10.1021/acs.est.1c05509
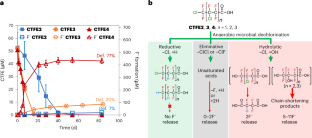
嫌気性微生物の加水分解によるポリクロロフルオロカルボン酸の大幅な脱塩素化 Substantial defluorination of polychlorofluorocarboxylic acids triggered by anaerobic microbial hydrolytic dechlorination
Bosen Jin,Huaqing Liu,Shun Che,Jinyu Gao,Yaochun Yu,Jinyong Liu & Yujie Men
Nature Water Published:15 May 2023
DOI:https://doi.org/10.1038/s44221-023-00077-6
Abstract
Chlorinated polyfluorocarboxylic acids (Cl-PFCAs) derived from the widely used chlorotrifluoroethylene oligomers and polymers may enter and influence the aquatic environment. Here we report the substantial defluorination of Cl-PFCAs by an anaerobic microbial community via novel pathways triggered by anaerobic microbial dechlorination. Cl-PFCAs first underwent microbial reductive, hydrolytic and eliminative dechlorination, with the hydrolytic dechlorination leading to the highest spontaneous defluorination. Hydrolytic dechlorination was favoured with increased Cl substitutions. An isolated, highly enriched, anaerobic defluorinating culture was dominated by two genomes that were most similar to those of Desulfovibrio aminophilus and Sporomusa sphaeroides, both of which exhibited defluorination activity towards chlorotrifluoroethylene tetramer acid. The results imply that anaerobic non-respiratory hydrolytic dechlorination plays a critical role in the fate of chlorinated polyfluoro chemicals in natural and engineered water environments. The greatly enhanced biodegradability by Cl substitution also sheds light on the design of cost-effective treatment biotechnologies, as well as alternative polyfluoroalkyl substances that are readily biodegradable and less toxic.
嫌気性および好気性条件下における不飽和パーフルオロおよびポリフルオロカルボン酸の微生物による脱フッ素化: 構造特異性の研究 Microbial Defluorination of Unsaturated Per- and Polyfluorinated Carboxylic Acids under Anaerobic and Aerobic Conditions: A Structure Specificity Study
Yaochun Yu, Shun Che, Changxu Ren, Bosen Jin, Zhenyu Tian, Jinyong Liu, and Yujie Men
Environment Science & Technology Published:April 4, 2022
DOI:https://doi.org/10.1021/acs.est.1c05509

Abstract
The recently discovered microbial reductive defluorination of two C6 branched and unsaturated fluorinated carboxylic acids (FCAs) provided valuable insights into the environmental fate of per- and polyfluoroalkyl substances (PFASs) and potential bioremediation strategies. However, a systematic investigation is needed to further demonstrate the role of C═C double bonds in the biodegradability of unsaturated PFASs. Here, we examined the structure-biodegradability relationships of 13 FCAs, including nine commercially available unsaturated FCAs and four structurally similar saturated ones, in an anaerobic defluorinating enrichment and an activated sludge community. The anaerobic and aerobic transformation/defluorination pathways were elucidated. The results showed that under anaerobic conditions, the α,β-unsaturation is crucial for FCA biotransformation via reductive defluorination and/or hydrogenation pathways. With sp2 C–F bonds being substituted by C–H bonds, the reductive defluorination became less favorable than hydrogenation. Moreover, for the first time, we reported enhanced degradability and defluorination capability of specific unsaturated FCA structures with trifluoromethyl (-CF3) branches at the α/β-carbon. Such FCA structures can undergo anaerobic abiotic defluorination in the presence of reducing agents and significant aerobic microbial defluorination. Given the diverse applications and emerging concerns of fluorochemicals, this work not only advances the fundamental understanding of the fate of unsaturated PFASs in natural and engineered environments but also may provide insights into the design of readily degradable fluorinated alternatives to existing PFAS compounds.