2024-10-16 カロリンスカ研究所(KI)
<関連情報>
- https://news.ki.se/new-study-on-micrornas-could-lead-to-better-fertility-treatment
- https://www.nature.com/articles/s41467-024-52943-w
ヒト着床前発生における低分子ノンコーディングRNAのアトラス An atlas of small non-coding RNAs in human preimplantation development
Stewart J. Russell,Cheng Zhao,Savana Biondic,Karen Menezes,Michael Hagemann-Jensen,Clifford L. Librach & Sophie Petropoulos
Nature Communications Published:05 October 2024
DOI:https://doi.org/10.1038/s41467-024-52943-w
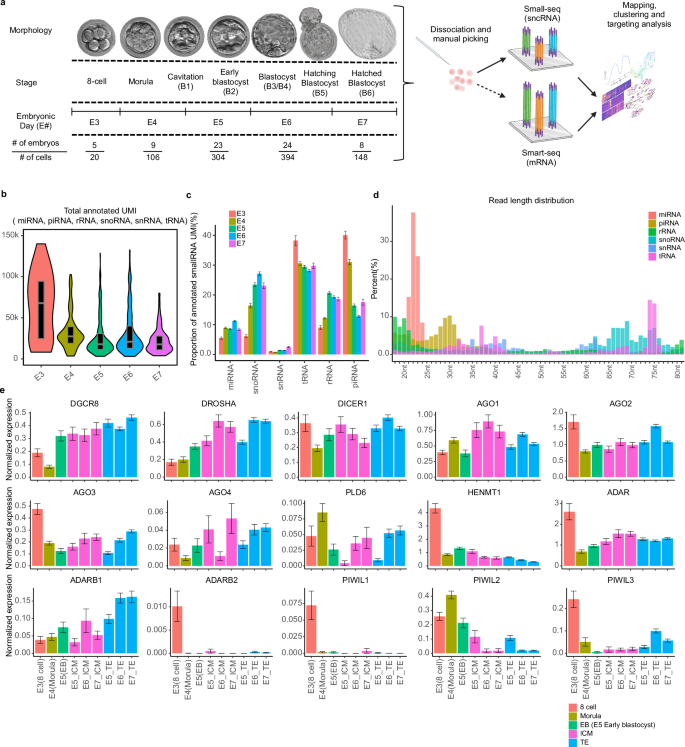
Abstract
Understanding the molecular circuitries that govern early embryogenesis is important, yet our knowledge of these in human preimplantation development remains limited. Small non-coding RNAs (sncRNAs) can regulate gene expression and thus impact blastocyst formation, however, the expression of specific biotypes and their dynamics during preimplantation development remains unknown. Here we identify the abundance of and kinetics of piRNA, rRNA, snoRNA, tRNA, and miRNA from embryonic day (E)3-7 and isolate specific miRNAs and snoRNAs of particular importance in blastocyst formation and pluripotency. These sncRNAs correspond to specific genomic hotspots: an enrichment of the chromosome 19 miRNA cluster (C19MC) in the trophectoderm (TE), and the chromosome 14 miRNA cluster (C14MC) and MEG8-related snoRNAs in the inner cell mass (ICM), which may serve as ‘master regulators’ of potency and lineage. Additionally, we observe a developmental transition with 21 isomiRs and in tRNA fragment (tRF) codon usage and identify two novel miRNAs. Our analysis provides a comprehensive measure of sncRNA biotypes and their corresponding dynamics throughout human preimplantation development, providing an extensive resource. Better understanding the sncRNA regulatory programmes in human embryogenesis will inform strategies to improve embryo development and outcomes of assisted reproductive technologies. We anticipate broad usage of our data as a resource for studies aimed at understanding embryogenesis, optimising stem cell-based models, assisted reproductive technology, and stem cell biology.