2025-04-01 東京大学
<関連情報>
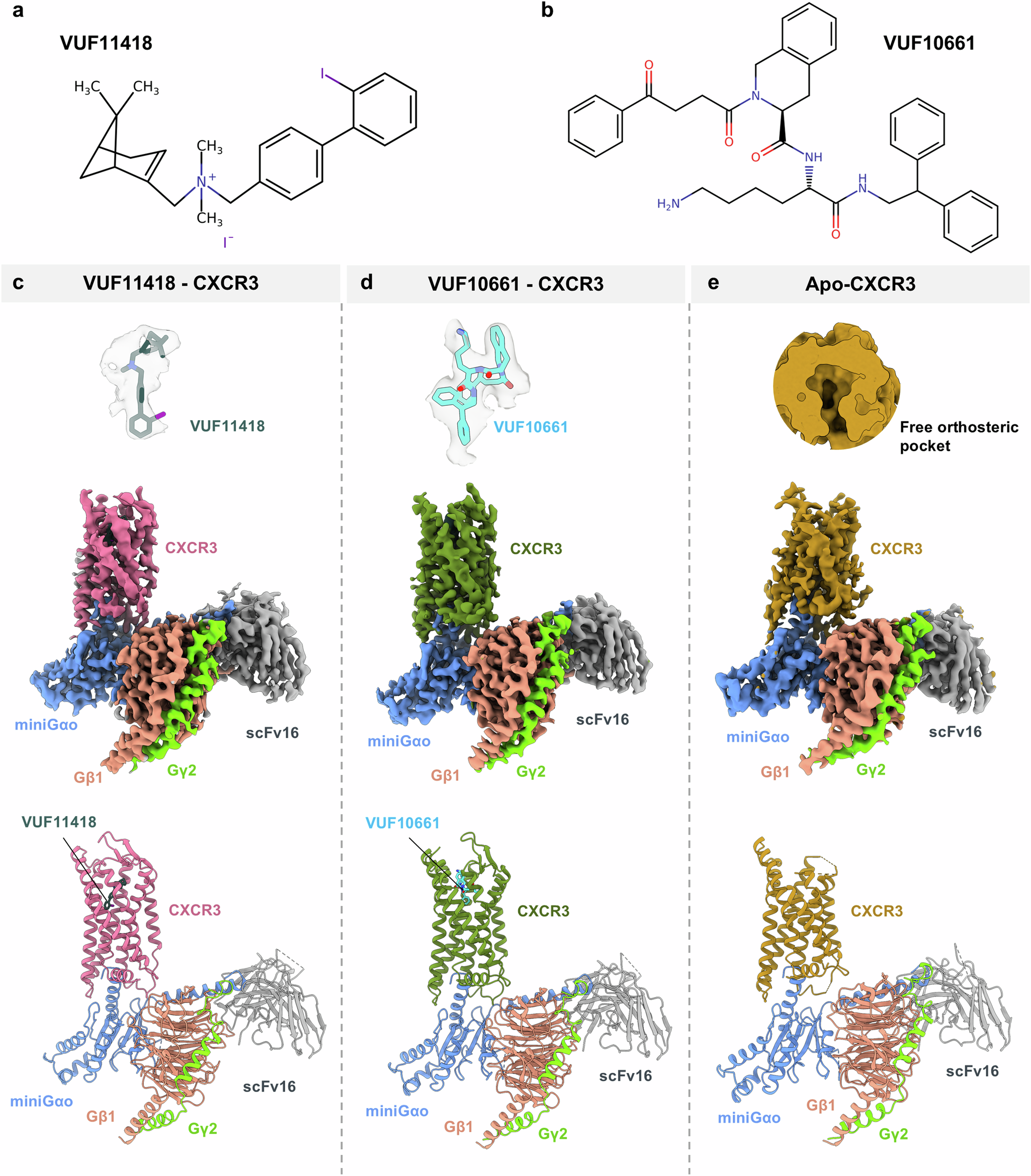
CXCR3による低分子認識の構造可視化により、CXCR3-CXCR7系における二重拮抗作用が明らかになった Structural visualization of small molecule recognition by CXCR3 uncovers dual-agonism in the CXCR3-CXCR7 system
Shirsha Saha,Fumiya K. Sano,Saloni Sharma,Manisankar Ganguly,Annu Dalal,Sudha Mishra,Divyanshu Tiwari,Hiroaki Akasaka,Takaaki A. Kobayashi,Nabarun Roy,Nashrah Zaidi,Yuzuru Itoh,Rob Leurs,Ramanuj Banerjee,Wataru Shihoya,Osamu Nureki & Arun K. Shukla
Nature Communications Published:28 March 2025
DOI:https://doi.org/10.1038/s41467-025-58264-w
Abstract
Chemokine receptors are critically involved in multiple physiological and pathophysiological processes related to immune response mechanisms. Most chemokine receptors are prototypical GPCRs although some also exhibit naturally-encoded signaling-bias toward β-arrestins (βarrs). C-X-C type chemokine receptors, namely CXCR3 and CXCR7, constitute a pair wherein the former is a prototypical GPCR while the latter exhibits selective coupling to βarrs despite sharing a common natural agonist: CXCL11. Moreover, CXCR3 and CXCR7 also recognize small molecule agonists suggesting a modular orthosteric ligand binding pocket. Here, we determine cryo-EM structures of CXCR3 in an Apo-state and in complex with small molecule agonists biased toward G-proteins or βarrs. These structural snapshots uncover an allosteric network bridging the ligand-binding pocket to intracellular side, driving the transducer-coupling bias at this receptor. Furthermore, structural topology of the orthosteric binding pocket also allows us to discover and validate that selected small molecule agonists of CXCR3 display robust agonism at CXCR7. Collectively, our study offers molecular insights into signaling-bias and dual agonism in the CXCR3-CXCR7 system with therapeutic implications.