2026-02-20 京都大学
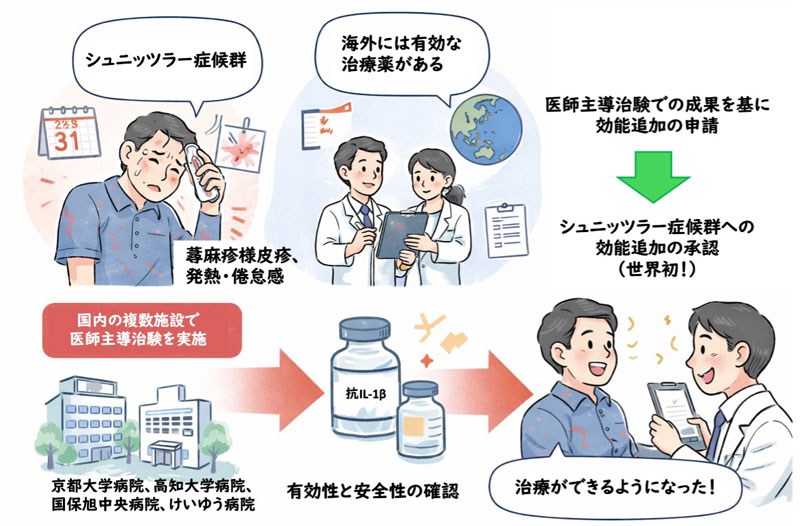
蕁麻疹様皮疹や発熱・倦怠感をきたすシュニッツラー症候群に対して、海外には有効な治療薬があるが、日本では使用できなかった。そこで、国内の複数施設で医師主導治験を実施し、その成果によって、世界初となる効能が追加された。(© 2026 Naotomo Kambe. Image created with ChatGPT 5.2)
<関連情報>
- https://www.kyoto-u.ac.jp/ja/research-news/2026-02-20
- https://www.kyoto-u.ac.jp/sites/default/files/2026-02/web_2602_Kambe-53a08bfb5192f6bda92c9b386dc70b04.pdf
- https://www.sciencedirect.com/science/article/pii/S1323893025000486
- https://www.sciencedirect.com/science/article/pii/S1323893024001175
シュニッツラー症候群では好中球がIL1B発現細胞として優位に働く:日本人患者におけるカナキヌマブの有効性と安全性を評価するSCan試験からの知見 Neutrophils predominate as IL1B-expressing cells in Schnitzler syndrome: Insights from the SCan study to evaluate the efficacy and safety of canakinumab in Japanese patients
Naotomo Kambe, Norimitsu Inoue, Yoko Ueki, Yuyi Zhou, Satoru Yonekura, Kosuke Katsuo, Satoshi Nakamizo, Hiroshi Tsujimoto, Katsuki Ohtani, Hajime Yoshifuji, Tomoyasu Jo, Kazushi Izawa, Mayuko Yamamoto, Koji Takemura, Shin-ichiro Kagami, Yoshie Kawahara, Yoko Amino, Yumiko Ibi, Satoshi Morita, Nobuo Kanazawa
Allergology International Available online: 19 May 2025
DOI:https://doi.org/10.1016/j.alit.2025.04.003
Abstract
Background
Schnitzler syndrome (SchS) is a late-onset autoinflammatory disease characterized by urticarial rash and monoclonal gammopathy. SchS shares clinical features with cryopyrin-associated periodic syndrome, which is driven by gain-of-function mutations in NLRP3, and while IL-1β-targeted therapies have shown efficacy, the underlying pathogenesis of SchS remains unclear.
Methods
During a multicenter, single-arm, open-label, investigator-initiated trial evaluating the efficacy and safety of canakinumab in five Japanese patients with SchS (named the SCan Study after SchS and Canakinumab), based on a similar study conducted in Germany, we measured 32 cytokines/chemokines and 11 complement-related factors in plasma and analyzed their correlations with changes in clinical symptoms during treatment. Furthermore, in two cases, single-cell RNA sequencing of peripheral blood and spatial transcriptomic analysis of lesional skin were performed to identify IL1B-expressing cells.
Results
The improvement in clinical symptoms and quality of life was maintained for 48 weeks following canakinumab treatment. Notably, these changes in clinical symptoms strongly correlated with WBC count, neutrophil count, CRP, and serum amyloid A levels, which were used as evaluation parameters in this study. In contrast, IL-1β and most other cytokines/chemokines exhibited distinct patterns and were not useful as markers of disease activity. IgM levels remained stable without an upward trend. Additionally, IL1B-expressing cells were predominantly neutrophils in both peripheral blood and lesional skin. Furthermore, neutrophil counts in peripheral blood decreased following canakinumab administration.
Conclusions
This study demonstrated that the primary source of IL1B-expressing cells in SchS is neutrophils. Moreover, canakinumab improves clinical symptoms by regulating neutrophil dynamics in peripheral blood.
シュニッツラー症候群の日本人患者におけるカナキヌマブの有効性に関する医師主導多施設共同単群オープンラベル試験 Investigator-initiated, multi-center, single-arm, open-label study of the effectiveness of canakinumab in Japanese patients with Schnitzler syndrome
Naotomo Kambe, Mayuko Yamamoto, Koji Takemura, Shin-ichiro Kagami, Yoshie Kawahara, Hajime Yoshifuji, Tomoyasu Jo, Kazushi Izawa, Satoshi Nakamizo, Norimitsu Inoue, Tatsuya Ito, Yoko Amino, Yumiko Ibi, Satoshi Morita, Nobuo Kanazawa
Allergology International Available online: 16 December 2024
DOI:https://doi.org/10.1016/j.alit.2024.10.001
Abstract
Background
Schnitzler syndrome is an adult-onset autoinflammatory disease characterized by an urticaria-like rash and monoclonal gammopathy with fever and fatigue. Although some treatments have shown efficacy in clinical trials, no approved treatment exists. We aimed to assess canakinumab, an anti-IL-1β monoclonal antibody, in Japanese patients.
Methods
This phase II, multicenter, single-arm, open-label study enrolled five patients with active disease from four hospitals. Patients received a single subcutaneous dose of canakinumab 150 mg. The primary endpoint was the proportion of patients achieving a complete clinical response (CR), based on physician global assessment on Day 7. If a CR was not achieved on Day 7 or by 8 weeks post-treatment, the dose was increased to 300 mg. Dosing continued every 8 weeks until 24 weeks. The study also evaluated patient-reported disease activity and changes in acute inflammatory markers, including white blood cell count, neutrophil count, C-reactive protein concentration, and serum amyloid A level. Quality of life was assessed using the Dermatology Life Quality Index and the 36-item Short Form health survey. Safety was also evaluated.
Results
Sixty percent (3/5) of patients had a CR on Day 7. One of the remaining two patients had a CR 7 days after the dose was increased to 300 mg. All five patients, including those who did not achieve a CR, showed improvement in inflammatory markers and quality of life scores, and no new adverse events were detected.
Conclusions
In this trial, canakinumab showed a potential for usefulness in Japanese patients with Schnitzler syndrome.