2026-02-25 東北大学
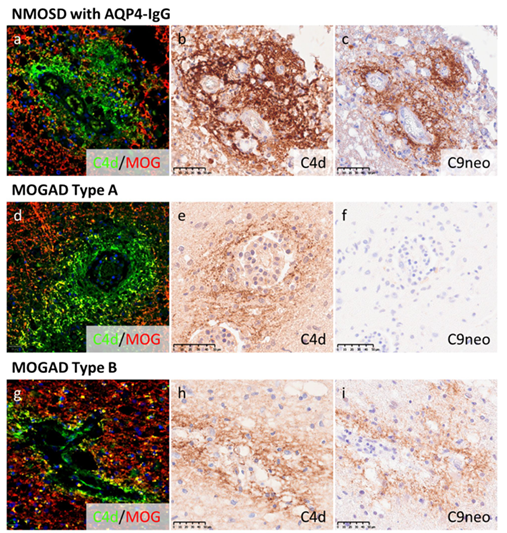
図1. NMOSDおよびMOGADにおける補体沈着パターン
<関連情報>
- https://www.tohoku.ac.jp/japanese/2026/02/press20260225-02-NMOSD.html
- https://www.tohoku.ac.jp/japanese/newimg/pressimg/tohokuuniv-press20260225_02web_NMOSD.pdf
- https://link.springer.com/article/10.1007/s00401-026-02985-9
NMOSD、MOGAD、MSにおける補体沈着の特徴的なパターン Characteristic patterns of complement deposition in NMOSD, MOGAD, and MS
Yoshiki Takai,Simon Hametner,Christian Riedl,Tatsuro Misu,Toshiyuki Takahashi,Hiroyoshi Suzuki,Norio Chihara,Masashi Watanabe,Hiroaki Miyahara,Mari Yoshida,Yasushi Iwasaki,Takashi Suzuki,Franziska Di Pauli,Stephan Bramow,Guy Laureys,Brenda Banwell,Sara Mariotto,Kazuo Fujihara,Masashi Aoki,Monika Bradl,Hans Lassmann & Romana Höftberger
Acta Neuropathologica Published:09 February 2026
DOI:https://doi.org/10.1007/s00401-026-02985-9
Abstract
The complement system is involved in the pathogenesis of inflammatory demyelinating diseases (IDDs) of the CNS. While complement inhibition significantly reduces the relapse rate in neuromyelitis optica spectrum disorders (NMOSDs), no clear consensus has been reached regarding the role of complement in myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) and multiple sclerosis (MS). Therefore, we examined CNS tissues from patients with NMOSD (18 autopsies and one biopsy, median age: 56 years), MOGAD (seven autopsies and 20 biopsies, median age: 34 years) and MS (24 autopsies, median age: 54.5 years) to assess the involvement of the complement system from a histopathological perspective. To investigate complement activity at multiple steps, the tissue deposition of three different complement components (C4d, C3d, and C9neo) was examined using immunohistochemistry. In NMOSD, the typical perivascular rosette/rim pattern of complement deposition was confirmed by the three different complement products within acute astrocyte-lytic lesions. In MOGAD, we observed C4d deposition around perivenous demyelinating lesions in 83% (20/24 tissues). However, C9neo deposition differed between patients, with 73% (11/15 patients with perivenous demyelination-predominant MOGAD) showing limited deposition of C9neo with relatively well-preserved oligodendrocytes (MOGAD type A), while 27% showing strong deposition accompanied by the disappearance of oligodendrocytes (MOGAD type B). The more destructive type B pathology was more frequent among deceased than living patients who, by contrast, had type A pathology in the vast majority. In MS, only C4d showed clear deposits on myelin sheaths in the peri-plaque white matter bordering the edges of the demyelinating lesions. These findings seemed to be characteristic of MS, and the extent and intensity tended to decrease in accordance with lesion activity. Complement deposition in MS lesions was linked to shorter interval between onset and death. These characteristic patterns of complement deposition in the three IDDs likely reflect the distinct pathogeneses of the diseases.