2026-03-05 東北大学
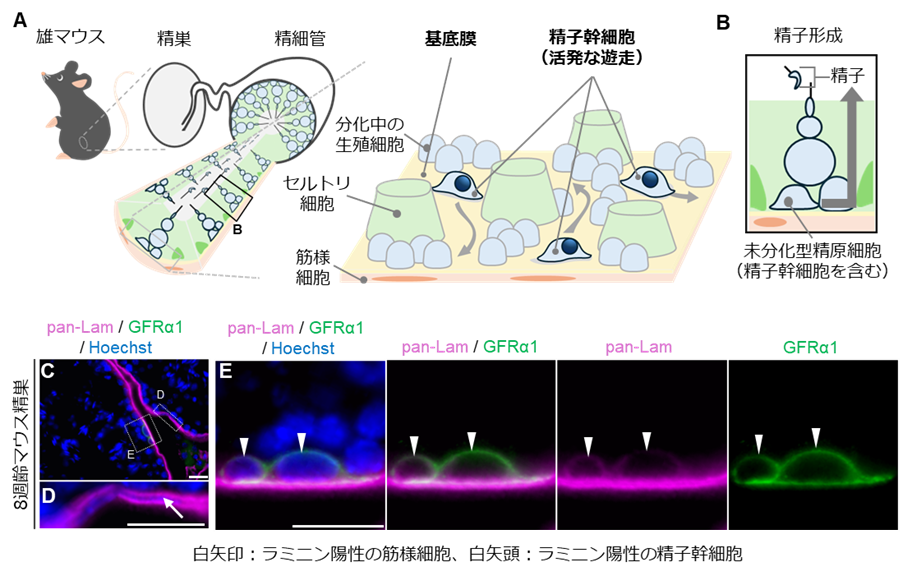
図1. マウス精巣における精子幹細胞の局在とラミニン発現。
<関連情報>
- https://www.tohoku.ac.jp/japanese/2026/03/press20260305-02-protein.html
- https://www.tohoku.ac.jp/japanese/newimg/pressimg/tohokuuniv-press20250305_02web_Protein.pdf
- https://academic.oup.com/biolreprod/advance-article/doi/10.1093/biolre/ioag032/8456225?login=false
未分化精原細胞は基底膜タンパク質ラミニンの発現を介してその行動を調節する Undifferentiated spermatogonia modulate their behavior via the expression of basement membrane protein laminin
Yusuke Kawabe ,Saya Yamada ,Yuichi Shima ,Kentaro Tanemura ,Shosei Yoshida ,Kenshiro Hara
Biology of Reproduction Published:03 February 2026
DOI:https://doi.org/10.1093/biolre/ioag032
Abstract
In the mouse testis, spermatogonial stem cells (SSCs) are sparsely distributed and migrate along the basement membrane of seminiferous tubules. Although the basement membrane is generally thought to be formed by surrounding somatic cells, whether SSCs also produce basement membrane proteins and, if so, whether SSC-produced laminin affects SSC behavior remains unknown. In this study, we found that mouse GFRα1+ spermatogonia, which include SSCs, expressed several laminin subunit genes, including Lamc1, whose expression declined upon differentiation. To test whether GFRα1+ spermatogonia-derived laminin regulates their behavior, we used two conditional knockout mouse models. In the Vasa-Cre model, which induces recombination in all germ cells, heterozygous deletion of Lamc1 increased both cell death and proliferation of GFRα1+ spermatogonia, while maintaining an apparent steady state of GFRα1+ cell density and spermatogenesis. In the tamoxifen-inducible GFRα1-CreER model carrying Lamc1flox/flox, tamoxifen-induced Lamc1 deletion in GFRα1+ spermatogonia caused a rapid reduction in their cell density within a few days, followed by increased proliferation and an imbalance between proliferation and differentiation of GFRα1+ spermatogonia that led to the restoration of GFRα1+ spermatogonial density. Collectively, these genetic findings suggest that GFRα1+ spermatogonia modulate their survival and behavior through laminin expression, likely by influencing the basement membrane around GFRα1+ spermatogonia. Such cell-autonomous regulation allows GFRα1+ spermatogonia, including SSCs, to form an appropriate local microenvironment wherever they reside within the testicular open niche, supporting stable behavior of spermatogonia during spermatogenesis.