2026-03-12 ミュンヘン大学(LMU)
<関連情報>
- https://www.lmu.de/en/newsroom/news-overview/news/how-flexible-protein-regions-retain-their-function-f5c7f6ec.html
- https://www.nature.com/articles/s41556-025-01867-8
配列と化学特異性が本質的に無秩序な領域の機能的景観を定義する Sequence and chemical specificity define the functional landscape of intrinsically disordered regions
Iris Langstein-Skora,Andrea Schmid,Frauke Huth,Drin Shabani,Lorenz Spechtenhauser,Mariia Likhodeeva,Franziska Kunert,Felix J. Metzner,Ryan J. Emenecker,Mary O. Richardson,Wasim Aftab,Maximilian J. Götz,Sarah K. Payer,Niccoló Pietrantoni,Valentina Valka,Sakthi K. Ravichandran,Till Bartke,Karl-Peter Hopfner,Ulrich Gerland,Philipp Korber & Alex S. Holehouse
Nature Cell Biology Published:12 February 2026
DOI:https://doi.org/10.1038/s41556-025-01867-8
Abstract
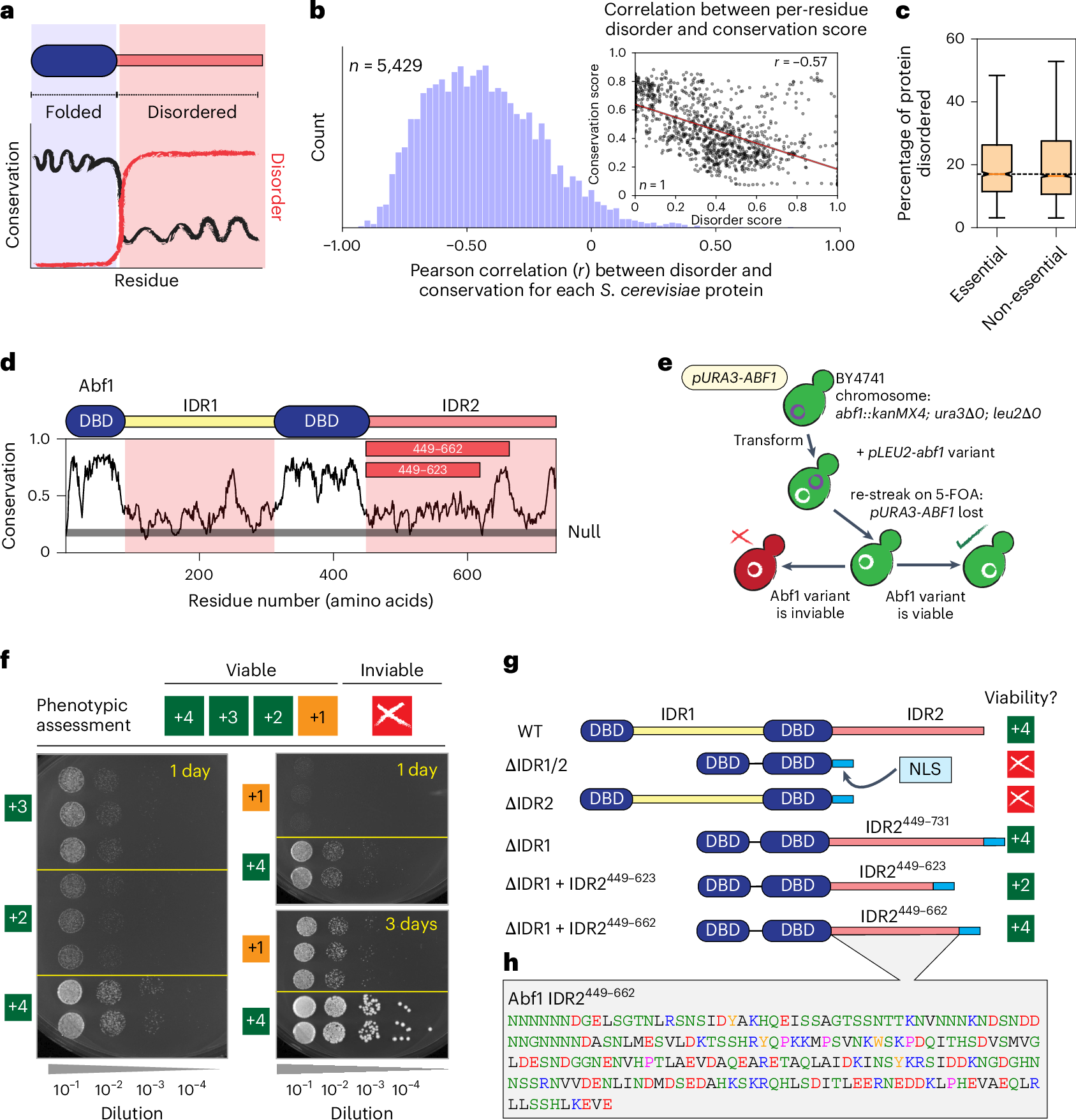
Intrinsically disordered regions (IDRs) pervasively engage in essential molecular functions, yet they are often poorly conserved as assessed by sequence alignment. To explore the seeming paradox of how sequence variability is compatible with persistent function, we examined the functional determinants for a poorly conserved but essential IDR. We show that IDR function depends on two distinct but related properties: sequence and chemical specificity. Whereas sequence specificity operates via binding motifs and depends on the precise order and identity of residues, chemical specificity reflects the sequence-encoded chemistry of multivalent interactions across an IDR and depends on local and global chemical properties. Unexpectedly, a binding motif essential in the wild-type IDR can be removed when compensatory changes to the sequence chemistry are introduced, highlighting the orthogonality and interoperability of these properties, and expanding the sequence space compatible with function. Our results provide a general framework for the functional constraints on IDR evolution.