2026-03-17 東京科学大学
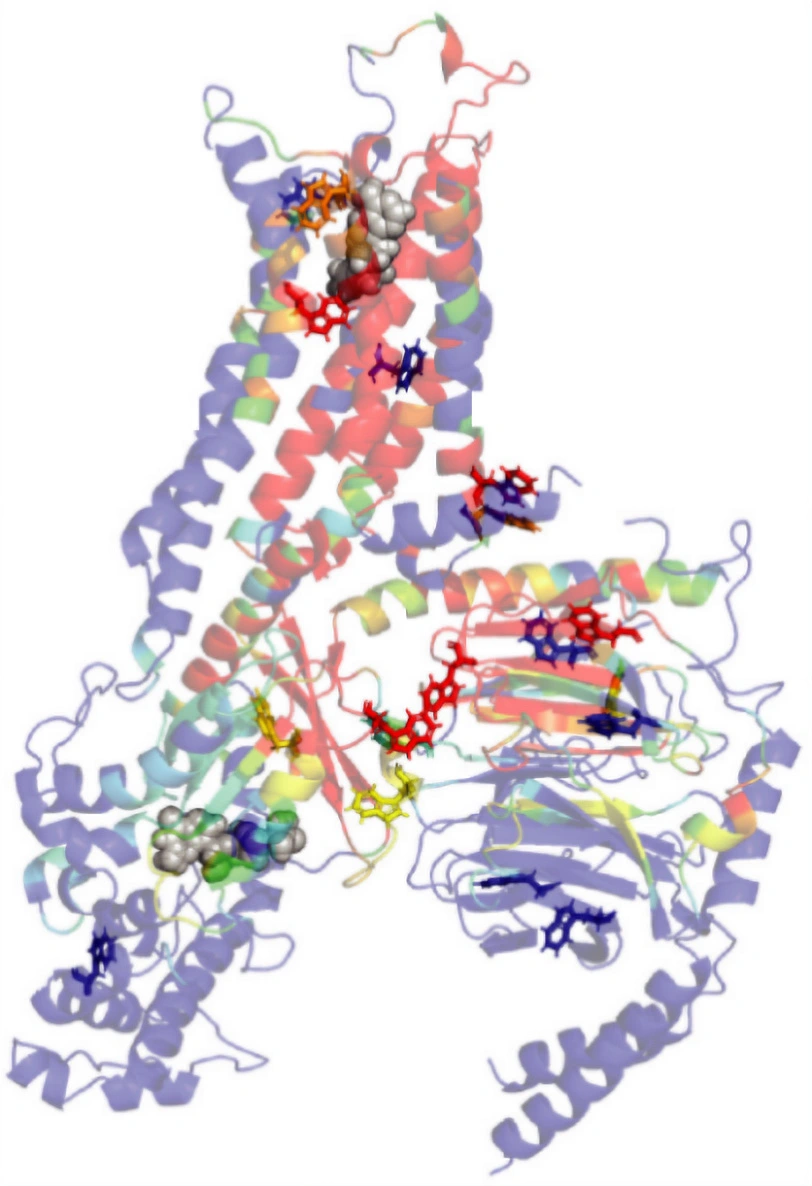
図1. GPCRにおけるトリプトファンに富むアロステリック・ネットワーク。
<関連情報>
GPCR活性化におけるトリプトファンに富むアロステリックネットワークとナトリウム排出の役割 The role of the tryptophan-rich allosteric network and sodium egress in GPCR activation
Zhenzhou Qi, Duy Phuoc Tran, Andrejs Tučs, +6 , and Robert Scott Prosser
Proceedings of the National Academy of Sciences Published:March 3, 2026
DOI:https://doi.org/10.1073/pnas.2525502123
Significance
While hundreds of class A GPCRs retain a sodium binding site in their inactive states, we have yet to explain why this is so crucial in receptor function. By monitoring both TM6 and a toggle switch adjacent to the sodium pocket, we can observe both toggle switch dynamics and the formation of a precoupled state which becomes stabilized as sodium egresses. This is likely a critical precursor to the onset of G protein coupling. Remarkably, tryptophan residues for the most part play a crucial role in GPCR activation, as the majority lie along computationally derived allosteric pathways. This allostery is explored in detail for A2AR using rigidity-theory and microsecond timescale molecular dynamic simulations.
Abstract
The human adenosine A2A receptor (A2AR) is a prototypical member of the class A family of G Protein–Coupled Receptors (GPCRs), which are engaged by over one-third of FDA-approved drugs. Here, we used 19F NMR to simultaneously evaluate functional states from the perspective of a CF3-tag on transmembrane helix-6 (TM6) and all 5-fluorotryptophan reporters. Whereas spectra from the TM6 tag reveal a dynamic conformational ensemble, the tryptophan spectra exhibit more discrete ligand-dependent states. These ligand-dependent signatures provide insights into microswitches including a universal toggle switch which interfaces with a sodium binding pocket. Over 560 GPCRs share this toggle switch — sodium pocket cluster, implying a fundamental role in activation. Computational rigidity-theory reveals tryptophan reporters reside along prominent allosteric activation pathways, underscoring their roles in mechanical signal propagation and corroborating NMR observations. Among them, W2466.48, associated with the toggle switch, plays a critical role in regulation of allosteric networks spanning the orthosteric pocket and extending via tryptophan-rich pathways through the receptor to the A2AR-Gβ interface and the nucleotide pocket in Gsα. While higher sodium concentrations (~100 mM) predictably stabilize the inactive conformation of the receptor, lower concentrations (below 40 mM) greatly enhanced the presence of the activation ensemble and in particular, the precoupled state, suggesting that the release of sodium from the conserved pocket—either through basal conditions for the apo receptor, or upon binding of the agonist—enables sampling of a precoupled state, ultimately needed to initiate activation and coupling.